Physical Pharmaceutics 2 - Unit 5
Syllabus
Drug stability: Reaction kinetics: zero, pseudo-zero, first & second order, units of basic rate constants, determination of reaction order. Physical and chemical factors influencing the chemical degradation of pharmaceutical product: temperature, solvent, ionic strength, dielectric constant, specific & general acid base catalysis, Simple numerical problems. Stabilization of medicinal agents against common reactions like hydrolysis & oxidation. Accelerated stability testing in expiration dating of pharmaceutical dosage forms. Photolytic degradation and its prevention
Scroll to Download
PHYSICAL PHARMACEUTICS-2 UNIT 5TH
DRUG STABILITY
Most Important Question
Drug Stability
- It is the ability of a drug to maintain its quality, potency and safety over time without degradation, when stored under specified conditions.
Reaction Kinetics
It includes the study of the speed or rate of chemical process that occurs during chemical reactions.
A (Reactant) B (product)
Rate of reactions, is the speed at which chemical reactions take place depends on concentration and time.
ZERO ORDER REACTIONS
These are those reactions, in which rate of reaction is independent of the concentration of the reactant.
In this, reaction rate remains constant because of zero order reaction.
- Let,
A B $$Rate = k[A]^x$$ At this, so,
- Rate of reaction
- So,
Where,
change in concentration with respect to time
specific rate constant for zero order.
Derivation :
The rate of zero order or
On integrating both sides,

Where
rate constant
starting concn of reactant at
concn of reaction (final) after time
Half Life : It is the time required to reduce half amount of Initial concentration of reactant.
Initial
Final at half time
- By putting these value in main equation -
- Half life is directly proportional to the initial concn of reactant.
Shelf Life : It is the time required to reduces the reactant concentration up to 90% from the initial concentration.
- Initial
- Final
Putting these value into main eq
Shelf life for zero order reactions
FIRST ORDER REACTIONS
It is, when rate of reaction depend on the concentration of reactant.
A B (product)
Equate both
or
Integration on Both Sides
For half life
Hint: in exponential form
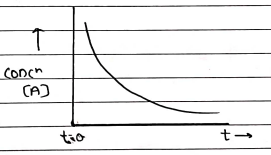
For shelf life (t_{0.9}$) $\rightarrow
Accelerated Stability Testing
- Stability of drug product is the ability of drugs to maintain its quality (retains its properties).
Expiry Date
It is the time period, at which drug is safe but after this time drug is not considered for use.
Now, expiration date of drug is selected on the basis of its shelf life and its accelerated stability testing.
Shelf Life
- It is defined as the time required for concn of the reactant to reduce 90% of its Initial / original concn.
- Is the period of time at which food/drug maintain its safety and quality.
Accelerated Stability Testing
- In this test, the rate of decomposition of drug is accelerated or increases by providing high temp, high humidity & light conditions.
- Because, high temperature increase instability or decomposition of drug products.
- From this, we get idea regarding decomposition of drug when it stored for long period of time.
- In this we rise the temp for storage.
- 25°C product
- 45°C product
- 40°C
- 60°C
- Relative humidity also increases
- It is based on Arrhenius equation. $$k = A e^{-E_a/RT}$$
- According to Arrhenius, for every 10°C rise in temp, speed of reaction increases upto 2-3 times.
- Product stored at diff-diff temperatures.

- After all this, Calculate its shelf life which is safe for the use of the products.
Degradation : It is the process of decreasing the quality of products.
Factor Affecting
- Temperature
- Light
- Moisture/humidity
- Degradation depends on pH, solvent, excipients, buffer.
Photolytic Degradation :
- It is the process of decreasing the quality of product through sunlight/light.
- Photon + lytic light breakdown its prevent Chem dy

