Physical Pharmaceutics 2 - Unit 3
Syllabus
Coarse dispersion: Suspension, interfacial properties of suspended particles, settling in suspensions, formulation of flocculated and deflocculated suspensions. Emulsions and theories of emulsification, microemulsion and multiple emulsions; Stability of emulsions, preservation of emulsions, rheological properties of emulsions and emulsion formulation by HLB method.
Scroll to Download
PHYSICAL PHARMACEUTICS 2ND UNIT-3RD
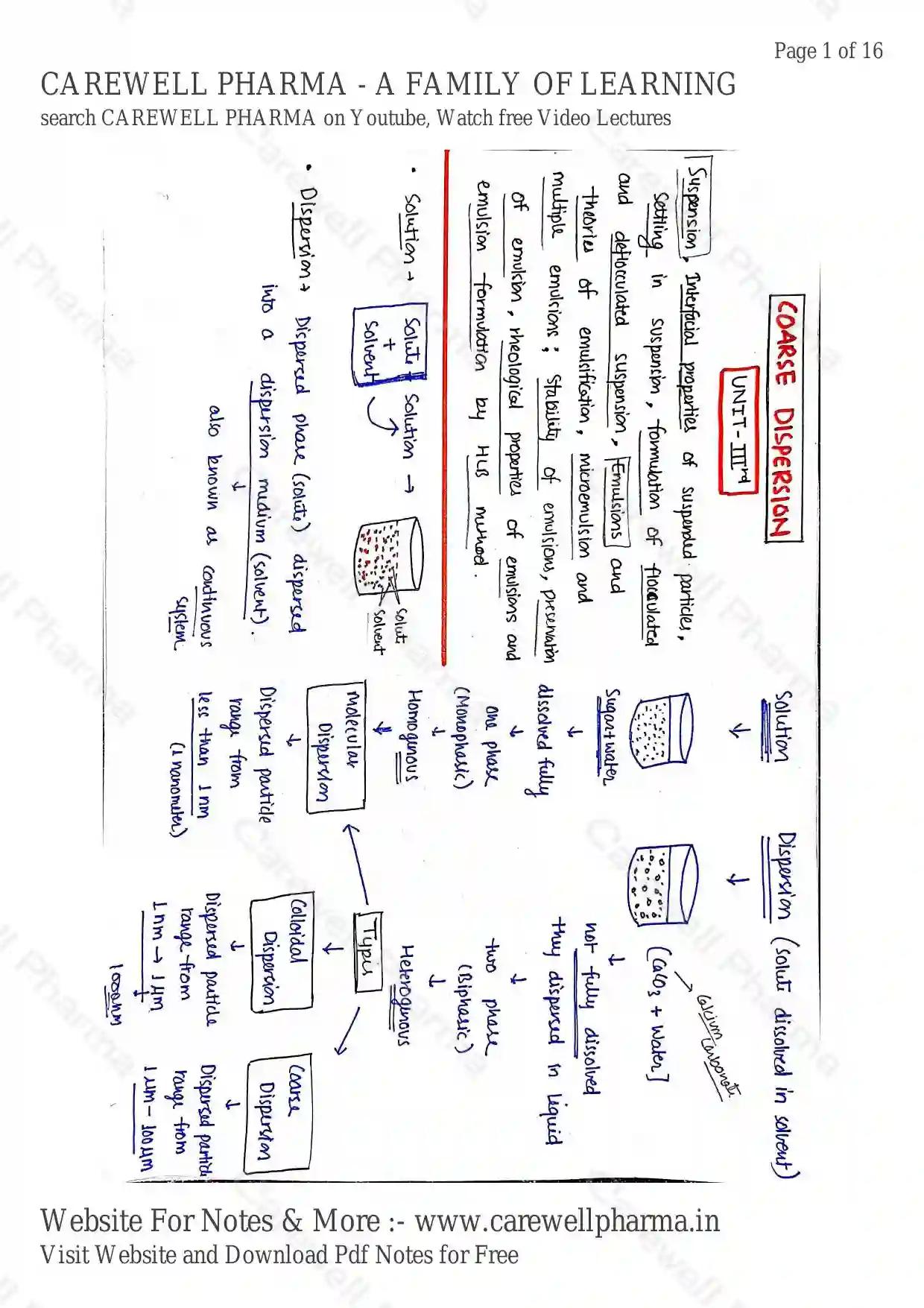
COARSE DISPERSION
Solution
- Solute + Solvent Solution one phase (Monophasic) Homogenous Molecular Dispersion Dispersed particle range from less than 1 nm (1 nanometer)
- Sugar + water dissolved fully
Dispersion (Solute Dissolved in Solvent)
- Dispersed phase (solute) dispersed into a dispersion medium (solvent). also known as continuous system.
- Calcium Carbonate [ + Water] not fully dissolved they dispersed in liquid two phase (Biphasic) Heterogenous
- Types:
- Colloidal Dispersion Dispersed particle range from 1 nm 1 / 1000 nm
- Coarse Dispersion Dispersed particle range from 1 - 100
Coarse Dispersion : It is a heterogenous dispersion system in which the size of dispersed phase particles range from 1 to 100 .
- Particles have size more than 1000 nanometer, so it can be easily visible with eyes.

Suspension is biphasic (both phase visible) and heterogenous in nature.
Suspension : It is a heterogenous system in which solid particles (dispersed phase) is uniformly dispersed in Liquid medium (dispersion medium).
- Dispersed phase solid (1 - 100 $\mu m$)
- Dispersion medium Liquid
- Eg. chalk in water etc-
- Suspension is biphasic (both phase visible) and heterogenous in nature.
Ideal Properties of Suspension
- Suspended molecules should not settle rapidly
- Prevent from cake formation at bottom.
- Easy to pour.
- Stable
- Easily flow out from syringe needle
Interfacial properties of suspended particles
In suspension, the solid particles is uniformly dispersed in Liquid medium. the Interface, is formed in between the two phases, which influence the stability of suspension.
i) Surface Free Energy :
During formulation of suspension, the solid is reduced to fine powder for better mixing in continuous medium. • Due to very fine particles, surface area of particles are increase and also their surface free energy increases.
surface free energy = Stability of suspension
Due to excess surface free energy, particles become highly energetics and tends to aggregates itselfs and resulting in formation of compact cake.
Equation
Where,
= Surface free energy.
= interfacial tension b/w solid & Liquid.
= Change in Surface area
- To reduce surface free energy and make suspension stable, we use wetting agents and surfactants.
- Also make the floccules to reduce the surface free energy and make suspension stable. eg. polysorbate-80 (wetting agents) etc-
ii) Formation of Electrical Double Layer :
- The ionic species present in solution get absorbed at the surface. Due to ionization charges on the surface may arise.
- This produce the formation of electric double layer.

- When surface charge is negative then their will be Layer of tightly bound solvent molecules which is stern layer, whereas the layer of positive charge, adjacent to the surface is called diffuse layer.
- Low electrolyte concentration deflocculated system
- More (increasing) electrolyte concentration flocculated system.
- Interaction (attraction) force b/w particles Flocculated
- Repulsive force b/w particles deflocculated

** Potential Energy curve for particle interaction.
Setting of Suspension
Also known as Rate of sedimentation, because settling of suspension is depend on sedimentation rate.
The settling down of suspended particles of suspension at bottom is known as settling of suspension or also sedimentation.
Rate of sedimentation = Rate of settling
The more rate of sedimentation, faster the particles settle down and faster the formation of cake.

The rate of settling of suspended particles is explained by stoke's law-
Where,
= Rate of sedimentation / Rate of settling
= diameter of particle
= density of suspended particle (dispersed phase)
= density of solvent (dispersion medium)
= gravity
= viscosity of dispersion medium.
- Rate of settling = Stability
- To make suspension stable :
- Increase viscosity of dispersion medium by using some agents such as methylcellulose, natural gums, acacia, tragacanth etc-) (structural vehicle method).
- Stoke's law is valid for those suspension which does not contain more than 2% solid.
Types of Suspension & formulation of flocculated and deflocculated suspension
Suspension Flocculated & Deflocculated
• On the basis of stability, suspension may be stable or unstable.

i) Flocculated : Flocculated suspension are those suspension in which particles of dispersed phase aggregates together to form bunch like structure called floccules.
ii) Deflocculated suspension : These are those in which particles of dispersed phase remain apart from each other and remain dispersed in the dispersion medium.

Flocculated • Rate of sedimentation is high. • more stable • form soft cake
Deflocculated • Rate of sedimentation is low • Less stable • form hard cake
- Flocculated are more stable than deflocculated:
Flocculated • It have large voids, so on setting down it form soft cake. • On Shaken, it redisperse condition. • More stable.
Deflocculated • It have less voids, so on setting down it form hard cake. • On shaken, its redispersion is very tough due to hard cake. • Less stable.
Formulation • Three various method which involved in formulation of suspension and increases its stability.
- Wetting Agent
- Controlled Flocculation
- Structure vehicle.
i) Wetting Agent
We known that less the particle size, less the rate of sedimentation but small particles size lead to more surface free energy that lead to formation of lymph, which is not a stable condition for suspension.
So, to tackle that we add wetting agent to the suspension that reduce the surface free energy and make suspension stable.
Eg: Polysorbate-80
ii) Controlled Flocculation :
- In this method, we add electrolytes to the suspension.
- When we add electrolyte OR charge develop on the particles and they repel each other. Hence make suspension stable.
- If they some how make the floccules, they will get easily redispersed.
- We will take the eletrolyte in such a way that it will be opposite charge for the solvent (dispersion medium)
iii) Structural Vehicle : In this method, we increase the viscosity of dispersion medium. So, that the particles (Light weight) does not sediment early in the suspension and remain dispersed. eg Acacia, Tragacanth etc-

EMULSION
It is a biphasic system of two immiscible Liquid in which one Liquid (dispersed phase) is dispersed into another Liquid (dispersion medium) with the help of any emulsifying agent.
Eg. Vanishing cream, Lotion, ointment etc-

Classification of Emulsion (Types)
It can be classified into four types:
i) Oil-in-water (O/w) emulsion
ii) Water-in-Oil (w/o) emulsion
iii) Multiple emulsion
iv) Microemulsion
i) o/w Emulsion : [aqueous emulsion]
- This are those emulsion in which dispersed phase is oil and continuous phase is water.
- Example : Milk, lotions, Liniments etc-- non-greasy nature (easily remove from body).
ii) w/o Emulsion : [oil emulsion]
- This are those emulsion in which dispersed phase is water and continuous phase is oil.
- Example : Butter and cold creams etc- Greasy in nature (oily nature, not easily removed from body).
iii) Multiple Emulsion : (Double Emulsion)
- Those emulsion which are made up of more than two phase.
- In which the droplets of one Liquid are dispersed in droplet of second layer Liquid, which is further dispersed in dispersion medium.
Types
O/W/O (oil-in-water-in-oil) : In which O/W emulsion dispersed in another oil phase
W/O/W (water-in-oil-in-water) : In which W/O emulsion dispersed in another water phase

- This types of emulsion prepared to prolong the release of drugs. • It enhance bioavailability.
iv) Micro Emulsion :
- Those emulsion in which particle size of dispersed phase is less than 1 (micron). It is clear, Transparent and thermodynamically stable as compared to normal emulsion.

Theories of Emulsion
Those theories which make and stay emulsion stable
i) Monomolecular theory
ii) Multimolecular theory
iii) Solid particle theory.
i) Monomolecular Adsorption Theory
- During preparation of emulsion, the oil/water droplet get dispersed in continuous phase, particle get atomized (reduce particle size).
- Due to small particle size surface area increases which further increases the surface free energy, therefore system become unstable.
- To resolve this problem or to make system stable we add surfactants or emulsifying agents which reduce the interfacial tension or surface free energy and make emulsion stable.
- Mechanism
- Surfactant contain polar and non-polar part. Polar oriented toward water and Non polar oriented toward oil and make emulsion. form a stable monomolecular film on the surface of dispersed droplet.

ii) Multimolecular Adsorption Theory :
- Monomolecular and multimolecular theory both used to stop flocculation, coalescence, also prevent the breaking of emulsion.
- Mechanism
- In this, emulsifying agent's small-small particles join together and cover the dispersed particles and Make multimolecular film.

- They also act by making the medium viscous.
Eg. Acacia, methyl cellulose etc-
• Flocculation Small particles forms a one big particles called floccules.
iii) Solid Particle Adsorption Theory :
- In this, we add solid emulsifying agent in the emulsion which increases viscosity of emulsion. $\downarrow$ Sedimentation & cracking
- It also prevent (reduce) coalescence of dispersed particles.
Eg. Colloidal clay etc...
iv) Formation of Electrical Double Layer :
- In emulsion, oil droplets contain either negative or positive charge.
- Due to present of charge they develop electrical double layer. (already discussed).
- Due to this oil droplets produce repulsion force into each other and they remain suspended.
- They prevent coalescence and breaking of emulsion.
Stability of Emulsion
- A stable emulsion is one in which the globules (dispersed phase) retain their size and remain uniformly distributed throughout the continuous phase as well as.

Some instability occur during storage and formulation of emulsion
i) Creaming
ii) Coalescence
iii) Breaking
iv) flocculation
v) phase inversion
vi) physical and chemical properties change
i) Creaming : It is a reversible phenomenan. In which droplets of dispersed phase come together or deposite at the surface of the emulsion.
(O/w) Upward Creaming
- In this, oil have low density so they come together at the surface of emulsion.
- Upword creaming.
(w/o) Downward Creaming
- In this, density of water (dispersed phase) is more than continuous phase.
- So particles come together at the bottom of emulsion.
- Downword Creaming.

ii) Coalescence : In emulsion, oily particles (dispersed phase) because of sticky nature attract each other and they aggregate to form big particles. oily particles aggregated.

iii) Breaking : (cracking)
- Due to improper mixing of oil and water in emulsion,
- Emulsion get separated into two layer i.e. oil and water.
- Is also occured due to improper ratio of oil and water.

iv) Flocculation : In this, due to increased surface free energy particle get aggregated to form floccules to decrease surface area.
v) Phase Inversion : In this, the phase of emulsion get inverted. i.e.. O/w w/o O/w
- It happened due to mixing problem or by choosing wrong dispersed phase during formulation.
vi) Physical & Chemical Property Change : To prepare an emulsion, we use emulsifying agent to mix and make stable emulsion.
- Sometimes, addition of these agents causes changes in properties like color, pH, odour, taste etc.
PRESERVATION OF EMULSIONS
- We have to keep emulsion stable and effective till expiry date. for this,
- We have to preserve emulsion from micro-organism and Oxidation.
- Emulsion (ideal property of preservatives)
- Not change their nature and property
- Not react oil and water with each other
- Stable
i) Preservation from Microorganism :
- Micro-organism may arrise in emulsion due to carbon content of emulsion or due to present of high water content.
- For this, we add Preservative Like methyl Paraben, propyl paraben, benzoic acid etc- which inhibit the growth of bacteria.
- Preservatives should be-
- Nonirritant and non-toxic
- Tasteless, colorless and odorless
- Should be stable
ii) Preservation from Oxidation
- Oxygen present in atmosphere cause Oxidative cause such as rancidity & spoilage.
- For this, we use some antioxidant which can prevent the changes occurs due to atmospheric Oxygen.
- Antioxidant should be
- Non-toxic and non-irritant.
- Effective at low concentration.
- Eg. BHT (Butylated Hydroxytoluene), Ethyl and propyl gallate etc
Rheological Properties of Emulsion
These properties of emulsion are related to the flow of emulsion.
Applications
- Spreadibility (for skin)
- Removal from container (bottle)
- Flow of Emulsion through hypodermic needle
- Proper mixing (during manufacturing)
- It can be of two types
1) Newtonian Flow : This type of flow show by dilute emulsion.
Eg. Micro-emulsion etc-
2) Non-newtonian Flow : This type of flow show by Concentrated emulsion.
Eg. Lotions, creams etc-
- Optimum viscosity is desirable for good stability of Emulsion.
- The increase in the viscosity of the emulsion reduce the flocculation.
- Rheological property of emulsion can be controlled by
- Concentration of dispersed phase.
- Particle size of dispersed phase
- Viscosity of continuous phase
- Nature and concentration of emulsifying system. etc_
FORMULATION OF EMULSION
- Method of preparation of emulsion
i) Dry Gum Method :
used for w/o type emulsion.
- Take a pestle and mortar
- Add gum to it (emulsifying agent powder form)
- Triturate it & then add some part of oil & triturate
- Again add some more oil and triturate untill receive the clicking sound.
- Then add water by part with trituration. Now make up the volume with bulk phase.
Ratio!- O:G:W = 4:1:2
ii) Wet Gum Method :
used for o/w type emulsion
- Take a Pestle and mortar.
- Add gum to it and triturate (emulsifying agent Liquid form)
- Add some water to gum and triturate with adding more water untill mucillage forms.
- Then add oily part slowly-slowly with Trituration.
- Now make up the volume with bulk phase. Ratio will be kept same as in i).
iii) Bottle Method : (Forbes Method)
- This method is used to prepare emulsions of volatile oils or substance having very low viscosity.
- 1 part gum placed in dry bottle then 2 parts of oil are added, Now shake the mixture.
- A volume of water (approx. equal to oil) is added in portion then again shake Now, Dilute it with proper volume of water.
- On large scale this process is done Inside bottle with or along with propeller movement.
Formulation by HLB Method
- High HLB Value showing emulsion emulsifying agent are mixed in O/w
- HLB Hydrophilic Lyophilic Balance
- With the help of HLB method we choose the emulsifying agent which we used in preparation of emulsions.
HLB SCALE
- Acc to HLB method,
- High HLB Value showing emulsifying agent are mixed in o/w emulsion.
- Low HLB value showing emulsifying agents are mixed in w/o emulsion.
- HLB scale decide to select emulsifier agents.
- More HLB value, more Hydrophilic (water soluble)
- Less HLB value, more Lipophilic (oil soluble)