Pharmaceutical Organic Chemistry 3 - Unit 5
Syllabus
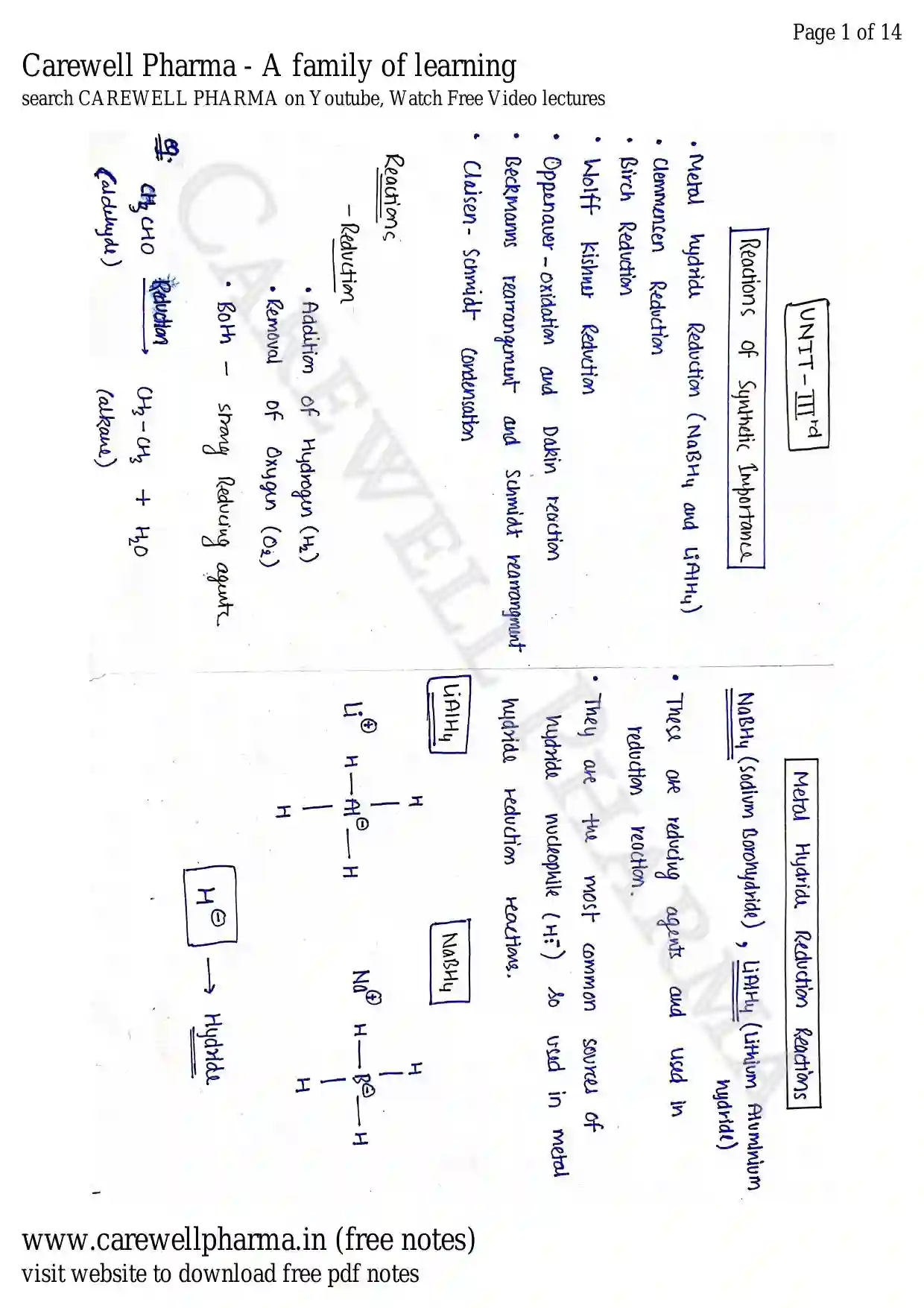
Reactions of synthetic importance
Metal hydride reduction (NaBH4 and LiAlH4), Clemmensen reduction, Birch reduction, Wolff Kishner reduction.
Oppenauer-oxidation and Dakin reaction.
Beckmanns rearrangement and Schmidt rearrangement.
Claisen-Schmidt condensation
Scroll to Download
POC-3rd UNIT-5th
Reactions of Synthetic Importance
- Metal hydride Reduction ($NaBH_4$ and $LiAlH_4$)
- Clemmencen Reduction
- Birch Reduction
- Wolff kishner Reduction
- Oppenaver - Oxidation and Dakin reaction
- Beckmanns rearrangement and Schmidt rearrangment
- Claisen - Schmidt Condensation
Reactions
Reduction
- Addition of Hydrogen ($H_2$)
- Removal of Oxygen ($O_2$)
- Both strong Reducing agents.
Eg :
(aldehyde) (alkane)
Metal Hydride Reduction Reactions
(Sodium Borohydride) , (Lithium Aluminium hydride)
- These are reducing agents and used in reduction reaction.
- They are the most common sources of hydride nucleophile ($H^\ominus$) so used in metal hydride reduction reactions.
[Image description: Chemical structures of and . shows a central Aluminum atom bonded to four Hydrogen atoms with a negative charge and a Lithium cation ($Li^\oplus$). shows a central Boron atom bonded to four Hydrogen atoms with a negative charge and a Sodium cation ($Na^\oplus$). An arrow points from to the word "Hydride"]
LiAlH_4$ (Lithium Aluminium Hydride)
It is a strong reducing agent.
It can reduce aldehyde to alcohols, ketones to alcohols.
Mechanism Two steps:
i) Nucleophilic attack by the hydride anion
ii) The alkoxide is protonated.
($2^\circ$ alcohol)
another example:
(Sodium Borohydride)
- It is also strong reducing agents but less reactive than .
- It can reduce aldehyde, ketones and acid chloride to alcohols.
eg: $$R-CHO \xrightarrow{\text{1) } NaBH_4 \text{ 2) } H_2O} R-CH_2-OH$$ (aldehyde) ($1^\circ$ alcohol)
eg: $CH_3CHO \xrightarrow{NaBH_4} CH_3-CH_2-OH$ (Acetaldehyde) (propanol)
- Mechanism
Step-1 $$R-\overset{O}{\overset{||}{C}}-H + Na^\oplus \overset{\ominus}{B}H_4 \rightarrow R-\overset{O^\ominus Na^\oplus}{\overset{|}{CH_2}} + BH_3$$
Step-2 $$R-\overset{O^\ominus Na^\oplus}{\overset{|}{CH_2}} \xrightarrow{H-OH} R-CH_2-OH + NaOH$$ ($1^\circ$ alcohol)
Another example
[Image description: Chemical reaction showing Benzaldehyde reacting with to yield Benzyl alcohol]
Clemmensen Reduction
It is a chemical reaction described as reduction of ketones (or aldehydes) to alkanes using zinc amalgam ($Zn(Hg)$) and Hydrochloric acid ($HCl$).
Aldehydes/Ketones Alkanes
(alkanes)
- Here, group of aldehydes and ketones is reduced to .
Reaction of Ketones.
(ketones) (Alkanes)
Mechanism
[Image description: Detailed mechanism of Clemmensen Reduction. The ketone reacts with Zinc ($Zn$) to form a radical anion, which takes up protons (2H^+$) to form a zinc-alcohol intermediate. This is followed by protonation ($H^+$), loss of a water molecule ($-H_2O$) forming a carbocation, and subsequent addition of electrons ($2e^-$) and protons ($H^+$) to yield the final alkane product $R-CH_2-R.]
Examples
I)
(ethanal) (ethane)
- When ethanol undergo reduction in the presence of zinc amalgam and HCl it give ethane
II) $$CH_3-\overset{O}{\overset{||}{C}}-CH_3 \xrightarrow{Zn(Hg), HCl} CH_3-CH_2-CH_3$$ (propanone) (propane)
III) $$CH_3-CH_2-\overset{O}{\overset{||}{C}}-H \xrightarrow{Zn(Hg), HCl} CH_3-CH_2-CH_3$$ (propanal) (propane)
iv)
[Image description: Chemical reaction showing Benzaldehyde reacting with to yield Toluene (Methylbenzene)]
BIRCH REDUCTION
The Reduction of Aromatic compounds in the presence of Metals ($Na, Li, K$) and alcohol in Liquid ammonia into a cyclohexadienes compounds.
[Image description: Benzene ring reacting with and to yield 1,4-Cyclohexadiene]
The mechanism begins with a single electron transfer from the metal to the aromatic rings forming a radical anion.
The anion then picks up a proton from the alcohol which results in a neural radical intermediate. Now same with other carbon, results the final product cyclohexadiene formed.
Ex.
[Image description: Naphthalene reacting with and to yield 1,4-dihydro naphthalene]
[Image description: Alkyl benzene reacting with and to yield a mixture of (2-Alkyl-1,4-cyclohexadiene) and (3-Alkyl-1,4-cyclohexadiene)]
[Image description: Birch reduction mechanism starting with a Benzene ring reacting with to form a radical anion. The radical anion abstracts a proton from to form a radical intermediate. An additional electron is transferred to form an anion, which abstracts another proton from to yield the final product, 1,4-cyclohexadiene.]
Wolff Kishner Reduction
The Reduction of aldehyde and ketones into alkanes in the presence of hydrazine and strong base.
- Reaction of aldehyde/ketone with hydrazine produce hydrazone.
- Reaction with a base to convert hydrozone to alkane.
Here, "$=O$" group of aldehyde & ketones is reduced to "$CH_2$".
Eg :
(Acetone) (Hydrazone) (Propane)
Mechanism
i) formation of hydrazone...
(Aldehyde/ketones) + (Hydrazine) (Hydrazone)
ii) formation of alkanes
Reaction with a base to convert hydrazone to alkane $$R-\overset{N-NH_2}{\overset{||}{C}}-R \xrightarrow{KOH, \Delta \text{ } 180^\circ C} R-\overset{H}{\overset{|}{C}}-R + N_2$$ (Hydrazone) (Alkane)
[Image description: Detailed step-by-step mechanism of the Wolff-Kishner Reduction converting a hydrazone to an alkane. It shows the base-catalyzed deprotonation of the hydrazone, resonance stabilization, protonation from water, a second base-catalyzed deprotonation leading to the expulsion of Nitrogen gas ($N_2$), and final protonation of the resulting carbanion to yield the alkane.]
- This rxn is same as clemmension, but it occured in the presence of hydrazine and strong base with heat. (difference from clemmension).
Another example
[Image description: Cyclopentanone reacting with followed by to yield Cyclopentane]
Synthesis of pyroles
[Image description: 2,5-hexanedione undergoing Wolff Kishner Reduction (WKR) to form 2,5-dimethylpyrrole]
Oppenaver - Oxidation Reaction
It is an organic reaction used to convert a primary or secondary alcohol to a ketone using another excess ketone reagents (acetone) and an aluminium tri-isopropoxide catalyst. $\hookrightarrow$ also used (butoxide).
[Image description: Chemical reaction showing a secondary alcohol ($2^\circ$ alcohol) reacting with Acetone in the presence of Aluminium Triisopropoxide where to yield a ketone and 2-propanol]
alcohol ketone [oxidation]
Acetone Alcohol [Reduction]
Mechanism
[Image description: Mechanism of Oppenauer Oxidation. i) The alcohol reacts with Aluminium Triisopropoxide to form an alkoxide-aluminum complex. ii) This complex coordinates with acetone forming a six-membered transition complex, transferring a hydride to yield the ketone and a new isopropoxide-aluminum complex. iii) The new complex reacts with water to release 2-propanol and regenerate the catalyst.]
Examples
[Image description: Four diverse structural examples of secondary alcohols undergoing Oppenauer Oxidation in the presence of and Acetone to yield their respective ketones, including unsaturated and polycyclic substrates.]
DAKIN REACTION
Dakin reaction is the replacement of the aldehyde group of ortho and parahydroxy and ortho amino benzaldehyde (or ketones) by a hydroxyl group (OH) on reaction with hydrogen peroxide ($H_2O_2$) in the presence of Base ($NaOH / Na_2CO_3$).
It is the replacement of aldehyde group by hydroxyl (OH) group in the presence of and Base
[Image description: Chemical reaction of o-hydroxybenzaldehyde reacting with and Base to yield Catechol]
Mechanism
S-1 - addition of nucleophile at the carbonyl carbon.
[Image description: Mechanism Step 1: Nucleophilic attack of the peroxide anion on the carbonyl carbon of the aldehyde.]
S-2 - On Rearrangement,
[Image description: Mechanism Step 2: The aryl ring migrates to the peroxide oxygen, cleaving the O-O bond to release a hydroxide ion, forming an ester intermediate.]
S-3 - Hydrolysis
[Image description: Mechanism Step 3: Base-catalyzed hydrolysis of the ester intermediate to yield Catechol and formic acid.]
Examples
[Image description: D-vanillin reacting with and to yield Pyrogallol-1-monomethyl ether]
[Image description: p-hydroxy acetophenone reacting with and to yield Hydroquinone]
- It is most commonly used to synthesized catechol ring.
- Also known as dakin oxidation.
- Synthesis of Hydroquinone.
Beckmanns Rearrangement Reactions
It is an organic reaction used to convert an oxime to an amide under acid catalyst by rearrangement.
Oxime
Amide
[Image description: Reaction of Ketoxime with Acid to yield an Amide] [Image description: Reaction of Aldoxime with Acid to yield an Amide]
- Mechanism
[Image description: Mechanism of the Beckmann Rearrangement. It shows the protonation of the oxime hydroxyl group, followed by the loss of water and simultaneous anti-migration of the alkyl/aryl group to the nitrogen, forming a nitrilium ion. The nitrilium ion is then attacked by water, losing a proton to form an imidic acid, which tautomerizes to the final amide.]
- Examples
i)
(Acetone oxime)
ii)
[Image description: Benzaldoxime reacting with to yield Benzonitrile by loss of $H_2O$]
iii)
[Image description: Methyl Phenyl ketoxime reacting with to yield n-phenyl acetamide]
- It is used in the synthesis of some drugs such as - Etazepine, Prozepine etc-
Schmidt Rearrangement
In this reactions, carboxylic acid, aldehyde or ketones is react with hydrazoic acid and gives amine or amide by rearrangement.
(carboxylic acid) + (Hydrazoic acid) amine
Mechanism
[Image description: Detailed mechanism of the Schmidt Rearrangement. The carboxylic acid is protonated, then attacked by hydrazoic acid. Following the loss of water, an intermediate undergoes rearrangement with the expulsion of nitrogen gas ($N_2$) to form an isocyanate. The isocyanate is subsequently hydrolyzed by water to release carbon dioxide ($CO_2$) and yield the primary amine.]
eg.
[Image description: Benzoic acid reacting with and to yield Aniline]
Claisen - Schmidt Condensation
- It is a reaction b/w two molecules of carbonyl compounds ($-C=O$), one of which should contain atleast two $\alpha$-hydrogen and also lacking of an alpha ($\alpha$) hydrogen.
Eg.
[Image description: Chemical reaction of Benzaldehyde with Acetophenone in the presence of to yield Benzalacetophenone]
(aldehyde) -Benzaldehyde + (ketone) -Acetophenone (Benzalacetophenone)
- Mechanism
- Enolate ion are formed compound heat with base (NaOH).
[Image description: Mechanism of the Claisen-Schmidt Condensation. Step 1: Base (NaOH) abstracts an alpha-proton from acetophenone to form an enolate ion. Step 2: The enolate ion acts as a nucleophile and attacks the carbonyl carbon of benzaldehyde. Step 3: Protonation of the resulting alkoxide by water. Step 4: Base-catalyzed dehydration (loss of $H_2O$) to form the final $\alpha,\beta$-unsaturated ketone, Benzalacetophenone.]