Pharmaceutical Organic Chemistry 3 - Unit 3
Syllabus
Heterocyclic compounds:
Nomenclature and classification
Synthesis, reactions and medicinal uses of following compounds/derivatives
Pyrrole, Furan, and Thiophene
Relative aromaticity and reactivity of Pyrrole, Furan and Thiophene
Scroll to Download
UNIT-3 PHARMACEUTICAL ORGANIC CHEMISTRY 3
HETEROCYCLIC COMPOUNDS
Syllabus
- Nomenclature and classification
- Synthesis, Reactions and Medicinal uses of following Compounds/derivatives: Pyrrole, furan, Thiophene
- Relative aromaticity and reactivity of Pyrrole, furan and Thiophene.

- But if we change atleast one carbon with any other netero atom, then this compound known as heterocyclic compounds.
Definition
"These are those cyclic compounds which contain atleast one different atom than carbon in a ring. OR "They are those organic compounds which contain ring or cyclic structure, in which it contain atleast two different atom."
- The atom present in cyclic ring other than Carbon is 'Hetero atom'.
- Most common heteroatom used are Nitrogen, Oxygen and Sulphur.
- Than others phosphorus, silica, Aluminium, Copper ete...
- Eg. Pyrrole, furan, Oxazole etc.....
- Most of the drug which are used in pharmaceutical science are heterocyclic compounds.
- These compounds may be aromatic and Non-arometic, acc. to their ring types
Hetero different Cyclic Ring/close
• It is cyclic • All are carbon So, this is HOMOCYCLIC • But If we change atleast one carbon with any other hetero atom, then this compound known as Heterocyclic compounds. These are those cyclic compounds which contain atleast one different atom than Carbon in a ring.
Syllabus
- Most of the drugs which are used in pharmaceutical science are Heterocyclic compounds.
- These compounds may be aromatic and Non-aromatic, acc. to their ring type.
Classification of Heterocyclic compounds
- This is acc (only) for B. Pharma 4th Sem - POC-III*
Heterocyclic Compound
- 5-membered heterocyclic compounds
- One heteroatom: eq. pyrrole
- More than one heteroatom: eq. Pyrazole, Imidazole
- 6-membered heterocyclic compounds
- One heteroatom: eg. pyridine
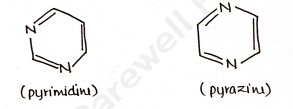
- more than one heteroatom: eg. pyrimidine
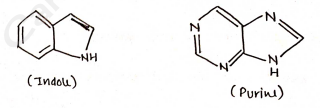
- Condensed heterocyclic compounds
- eq. Indole, Quinoline, Isoquinoline
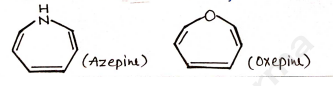
- 7-membered heterocyclic compounds
- eq. oxepine
- Heterocyclic compound classified as many ways!-
Acc. to their ring type it can classified as
- Aromatic - follow huckle rule.
- Non-aromatic not follow huckle rule
On the basis of members of ring
5-membered heterocyclic compounds
A Ring contains five atoms
- One hetero atom eg. (4 carbon + 1 heteroatom).

- More than one heteroatom eg.

6-Membered Heterocyclic Compound
Those heterocyclic compounds in which six atoms are present OR involved in ring.
One heteroatom eg. [5 Carbon + 1 heteroatom]
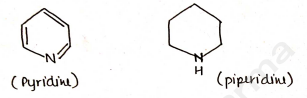
More than one heteroatom eg.
7-Membered Heterocyclic Compounds
A heterocyclic compounds contain 7 atom [6 Carbon + 1 heteroatom] eg.
Condensed Heterocyclic Compounds
Those heterocyclic compounds in which atleast two ring are fused.
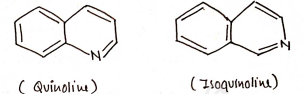
Nomenclature of Heterocyclic compounds
There are three system for naming heterocyclic compounds:-
- Common name system (trivial system of nomenclature)
- Replacement method
- Hantzsch widman method - IUPAC System
i) Common Name System
It is also known as a trivials name system.
These are the common name of a heterocyclic compounds which usually originated from the compounds occurence, its first preparation or its special properties.
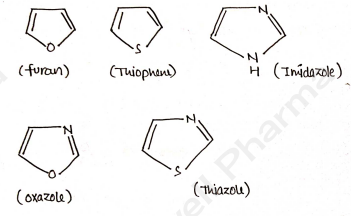
5-membered heterocycles
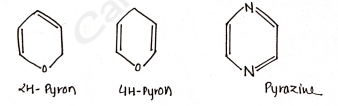
6-membered heterocycles
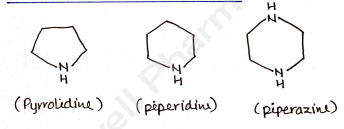
Saturated heterocyclic compounds
Fused heterocycles
ii) Replacement Nomenclature
Heterocycle's name is composed of the corresponding carbocycle's name and an elemental prefix for the heteroatom.
Example

iii) Hantzsch-Widman Nomenclature (IUPAC)
It is the most widely used system and also called as IUPAC nomenclature System.
- Proposed by Arthur Hantzsch and Oskar Widman in 1887 and 1888.
- IUPAC Prefix + Suffix
Some prefix of heteroatoms.
Oxygen (O) Oxa
Nitrogen (N) Aza
Sulphur (S) Thia
Phosphorus (P) Phospha
Silicon (Si) Sila
Bismuth (Bi) Bisma
- Suffix
| No. of membered | Saturated (-) | Unsaturated (=/$\equiv$) | ||
|---|---|---|---|---|
| with 'N' | without 'N' | With 'N' | Without 'N' | |
| 03 - ir | iridine | irane | irine | irene |
| 04 - et | etidine | etane | ete | et |
| 05 - ol | olidine | olane | ole | ole |
| 06 - in | ane | ine | in | |
| 07 - ep | epane | epine | epin | |
| 08 - oc | ocane |
Eg.
- Firstly choose heteroatom's prefix, then acc to Table choose the prefix.
- The terminal 'a' of prefix is removed when (on) combination of prefix and suffix.
- If there are more than one heteroatom are involved, then prefix are placed in order of priority O, S, Se, N, P, As, Si, B .... O > S > N.
Examples:

PYRROLE
- It is five membered heterocyclic compound containing nitrogen (N) as a hetero atom.
- Chemical formula
- Molecular weight 67.
- It occurs naturally in alkaloids, chlorophyll, haemoglobin etc...

Resonance
- Pyrrole is aromatic in nature.
- There are delocalisation of lone pair (present on N) and bond.
- Pyrrole show total five resonating structure.

Physical Properties
- Pyrrole is liquid which rapidly turns brown on exposure to air.
- Weakly basic in nature.
- Sparingly soluble in water but dissolve in ethanol and ether.
- Boiling point
Synthesis (Method of Preparation)
i) From Furan : By passing the mixture of furan, Ammonia and steam over the alumina (aluminium oxide catalyst) heated at .

ii) From Succinimide : Succinimide on distillation with zinc dust give pyrrole.

iii) From Acetylene : Mixture of acetylene and ammonia are pass through red-hot tube to yield pyrrole.

iv) Paal-Knorr Synthesis (from 1,4-diketones) : In this reaction, 1,4-diketone compound react with ammonia and gives derivatives of pyrrole.

Mechanism:

v) Knorr Pyrrole Synthesis : It involves the condensation of $\alpha$-amino ketone derivatives with a $\beta$-keto ester in the presence of acetic acid.

Chemical Reactions
i) Electrophilic Substitution Reaction : Pyrrole has lone pair on nitrogen which have ability to donate electron so, pyrrole is more reactive than benzene and easily give this reactions. (occurs at or 2nd position).
Nitration
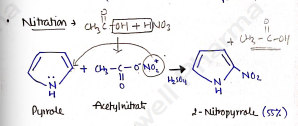
In this reaction, pyrrole is reacted with acetylnitrate in the presence of sulphuric acid to give 2-nitropyrrole.
Sulphonation

In this reaction, pyrrole is treated with Sulphuric acid in the presence of pyridine (agent) to gives pyrrole-2-sulphonic acid.
Friedel Craft Acylation

In this reaction, pyrrole is react with acetic anhydride to give 2-Acetylpyrrole.
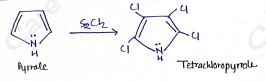
Chlorination
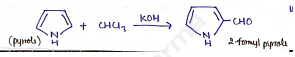
Riemer-Tiemann Reaction
ii) Reduction Reaction

- In this reaction, pyrrole react with zinc and acetic acid to give 3-pyrroline. But on react with hydrogen in presence of nickel give pyrrolidine.
iii) Oxidation reaction

- In the reaction, pyrrole is oxidised with chromium trioxide in the presence of acetic acid to give Maleinimide.
Medicinal use of Pyrrole & their derivatives
Pyrrole and its derivatives are widely used as an Intermediate in synthesis of pharmaceutical Medicines, agrochemicals, dyes, photographic chemicals, perfumes and other organic compounds.
Some derivatives and their medicinal uses
i) Procyclidine : It is an anti-muscarinic drug used in the treatment of parkinsonism.
ii) Atorvastatin: Useful to prevent cardiovascular disease.
iii) Prodigiosin: It has antibacterial, antifungal, anti-malaria and immunosuppressant activity.
iv) Triprolidine: used as antihistamines.
v) Elopiprazole: used as antipsychotic drug.
vi) Lincomycin, Clindamycin: used as antibiotics.
vii) Ondansetron: used as anti-emetic.
viii) Atoracetam: used in treatment of Alzheimer's disease.
FURAN
• It is five membered heterocyclic compounds containing oxygen (O) as a heteroatom.

- Chemical formula
- Molecular weight 68
- Other name Oxole
- Hybridization (carbon), (oxygen)
- Furan is aromatic in nature.
lone pair of oxygen, involved in delocalization.
- Orbital representation of furan.

Resonance

Physical Properties
- It is a colourless liquid.
- It is only slightly soluble in water.
- It has a chloroform like smell.
- Boiling point is .
Synthesis (method of preparation)
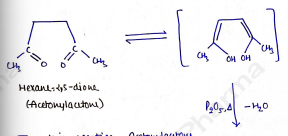
i) Paal Knorr Synthesis (from 1,4-diketo compounds)
In this reaction, Acetonylacetone (Hexane-2,5-dione) is heated with to form 2,5-dimethyl furan.
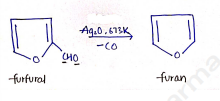
ii) From Furfural
In this reaction, furfural undergoes decarboxylation (remove CO) by heating in steam at 673K in the presence of silver oxide ($Ag_2O$).
iii) Feist-Benary Synthesis
- In this reaction, $\beta$-ketoester (Ethyl acetoacetate) react with $\alpha$-haloketones ($\alpha$-chloroacetone) in the presence of pyridine to gives derivative of furan.
iv) From Furoic Acid
- Decarboxylation of furoic acid yield furan.

v) From Pentose Sugar
- In this reaction, pentose sugar compound is dehydrated and formed furfural, which further undergoes decarboxylation and formed furan.

Chemical Reactions
Furan is an aromatic compound, so it give electrophilic substitution reaction.
i) Electrophilic Substitution Reaction :
- In acidic condition, furan get polymerized. So its reactions are carried out under mild reagents/conditions (occurs at or 2 position).
Nitration
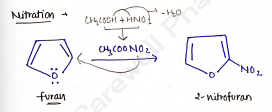
In this reaction, furan is treated with mixture of acetic anhydride and nitric acid to give 2-nitrofuran.
- Sulphonation

- In this reaction, furan is treated with pyridine-$SO_3$ complex to give 2-furan sulphonic acid.
- Friedel Craft Acylation

Furan on reaction with acetic anhydride in the presence of boron trifluoride gives 2-acetyl furan.
- Bromination

ii) Reduction (Hydrogenation)

- Furan on catalytic hydrogenation yields THF.
iii) Oxidation Reaction

- On oxidation, furan gives Succinaldehyde via formation of 1,4-peroxide.
Medicinal use of furan & their derivatives.
- Furan and their derivatives show many pharmacological action such as antidepressants, analgesic, muscle relaxant, antihypertensive etc...
- Used as solvent for resins.
- Used in the production of agriculture chemicals, stabilizers and insecticides.
Some drugs with their use!-
i) Prazosin: used as antihypertensive.
ii) Pilocarpine: used in treatment of open angle glaucoma.
iii) Ranitidine: inhibit stomach acid production.
iv) Lupitidine: antiulcer agent.
v) Nitrofurazone: used as an antibiotics.
THIOPHENE
- It is five membered heterocyclic compound containing Sulphur (S) as a heteroatom.

Discovered by Victor Meyer in 1882.
- Chemical formula
- Molecular weight 84
- Hybridization (4 Carbon + 1 sulphur) .
Thiophene is aromatic in nature

Resonance

Physical properties
- It is a colourless liquid.
- It is insoluble in water, but miscible with most organic solvent.
- It have an odour very similar that of benzene.
- Boiling point is .
Synthesis (Method of Preparation)
i) From Sodium Succinate (in Laboratory)

- In this reaction, thiophene is synthesized by heating sodium succinate with phosphorus trisulphide.
ii) From n-butane

- In this reaction, n-butane is heated with sulphur at and undergoes cyclization and formed thiophene.
iii) Paal Knorr Synthesis

- In this reaction, Acetonylacetone is heated with phosphorus pentasulphide and formed derivatives of pyrrole (2,5-dimethyl thiophene).
iv) From Acetylene

- In this reaction, mixture of acetylene and hydrogen sulphide is passed through a tube containing alumina at 673K.
Chemical Reactions
It is also a shows aromatic character but it is less reactive than pyrrole and furan.
1) Electrophilic Substitution Reaction
Thiophene can easily give this reactions (occurs at or 2-position).
- Nitration

- Sulphonation

- Friedel craft acylation

- Chlorination

ii) Reduction

- In this reaction, Thiophene reduced with Sodium in the presence of liq. ammonia give a mixture of 2-thiolen and 3-thiolen.

Medicinal Use of Thiophene
- Thiophene and its derivatives has many pharmaceutical importance (used as medicines).
- Used as anticancer agent (chemotherapeutic agents), diuretic, antiasthmatic agents etc...
- They have sedative, hypnotic, anticonvulsant and anxiolytic effect. clotiazepam.
- Some drugs:
i) Raltitrexed: anticancer activity.
ii) Cephalothin, Cefoxitin: antimicrobial agent.
iii) Tiamenidine: antihypertensive.
- Thiophene derivatives are also useful as pesticides.
Relative aromaticity and reactivity of pyrrole, furan and thiophene
The compound must be aromatic when it follow:
- cyclic
- must be in conjugation
- planar
- IV) follow Huckle's rule (4n+2) electron
Pyrrole, furan and thiophene all are aromatic in nature.
Relative Aromaticity
- Pyrrole contain 'N', Furan contain 'O', Thiophene contain 'S',
- Electronegativity O > N > S.
- E.N Delocalisation Aromatic Character Ar. Character S > N > O
- Acc. to this, Thiophene > Pyrrole > furan
- But all have low (less) resonance energy than benzene. So, less aromatic than benzene. Benzene > Thiophene > Pyrrole > furan <- Aromaticity
Relative Reactivity
Heterocyclic compound's resonance energy are much smaller than benzene hence they are more reactive.
- Relative Reactivity Pyrrole > furan > Thiophene > Benzene Which is more electronegative is more reactive. acc. to this, furan > pyrrole > thiophene (XX) But in case of furan, oxygen accommodates a positive charge are less reactive than nitrogen. So furan is less reactive than Pyrrole. (N is trivalent)
Order of reactivity: Pyrrole > furan > thiophene
- More Basic Pyrrole or Pyridine Pyridine > pyrrole <- Basic
- Lone pair donate more basic
- Resonance Stable less Basic
















