Pharmaceutical Organic Chemistry 3 - Unit 2
Syllabus
Geometrical isomerism
Nomenclature of geometrical isomers (Cis Trans, EZ, Syn Anti systems)
Methods of determination of configuration of geometrical isomers.
Conformational isomerism in Ethane, n-Butane and Cyclohexane. Stereo isomerism in biphenyl compounds (Atropisomerism) and conditions for optical activity.
Stereospecific and stereoselective reactions
Scroll to Download
POC-3 UNIT-2
GEOMETRICAL ISOMERISM
• The isomerism which arises due to restricted (frozen) rotation about double bond in molecules OR about single bond in cyclic compounds is known as geometrical or cis-trans isomerism.

• Geometrical isomerism show by variety of compounds such as -
- Compound containing double bond C=C , C=N , N=N
- Compound containing cyclic structure
- Restricted Rotation.
• Condition for geometrical isomerism -
i) There should be restricted (frozen) rotation about a bond in the molecules.
ii) Both substituents on each carbon about which rotation is frozen (restricted) should be different.

• Nomenclature of Geometrical Isomers [Cis-Trans, EZ, Syn Anti System]
Geometrical Isomers can be nomenclatured by three methods
- Cis Trans Nomenclature
- E Z nomenclature
- Syn Anti Nomenclature.
1) Cis Trans Nomenclature
Compounds show this nomenclature due to restricted rotation about carbon-carbon double bond.

- Compounds should have atleast one double bond b/w carbon carbon.
- Atleast one similar atom/group between both double bonded carbon.
- Restricted Rotation.
• Cis : The isomer in which the identical groups are on the same side of the double bond.
• Trans : The isomer in which the identical group are on the opposite sides of the double bond.
examples


2) E Z Nomenclature
This nomenclature of geometrical isomers is more general and can be applied to all compounds.
- Z (Zusammen together)
- E (Entegegen Opposite)
- It is based on Cahn-Ingold-Prelog system [Sequence Rule].
- The group of highest priority on each carbon atom is identified by using the sequence rules

(Z)-isomer (E)-isomer
- (Z) - If the highest priority group are on the same side of the double bond.
- (E) - If the highest priority group are on the opposite sides of the double bond.

3) Syn Anti Nomenclature
This type of nomenclature (isomerism) show by those compounds which have atleast one C=N or N=N.
Syn Same
Anti Opposite- Syn - If H on Carbon and substitute on Nitrogen are on same side.
- Anti - When H on Carbon and substitute on Nitrogen are on opposite sides
Methods of determination of configuration of geometrical isomers
- There are a number of methods to determine the configuration of geometrical isomers.
- These are depending on the nature of the compounds.
i) Physical method
ii) Cyclisation method
iii) Conversion method
i) Physical Method
- The melting point and intensity of absorption of the cis-isomer are lower than those of the trans. Trans>Cis.
- The boiling point, solubility, heat of combustion, heat of hydrogenation, density, refractive index, dipole moments and dissociation constant of the cis-isomers are greater than those of the trans. Cis>Trans.
- So, By comparing these properties compound identified as cis or trans.
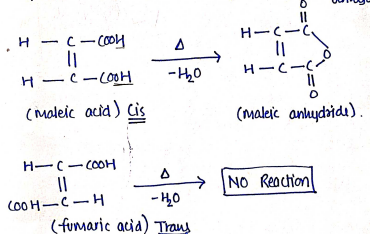
ii) Cyclisation Methods: (Methods of Cyclisation)
A cis isomers undergo cyclisation much more readily than the trans isomers.
Example : Maleic acid forms a cyclic anhydride (maleic anhydride) when heated, whereas fumaric acid does not form fumaric acid anhydride.
iii) Conversion Method: (method of conversion into compound of known configuration)
In a number of cases it is possible to determine the configuration of geometrical isomers by converting them into compounds of known configuration.
Example:

Conformational Isomerism in Ethane, n-butane and Cyclohexane
Conformational Isomers
These are different spatial arrangements of a molecules that are generated by rotation about single bonds.
- Use Newman Projection to these isomers.
- The structure of conformers is of two types
i) Staggered - Torsional angle is .
ii) Eclipsed - Torsional angle is .
Newman projection - Used to represent 3D structure.
Different conformations of the same molecules are sometimes called conformers/rotomer & conformational isomers.
Torsional angle - The angle between the atom attached to the front and rear carbon atoms. (Dihedral angle).
Examples - Ethane [$CH_3-CH_3$]
1) Ethane
When an ethane molecule rotates about its carbon-carbon single bond, two conformations :- staggered and eclipsed.
- Now, there are also use of energy during rotation.
- Eclipsed conformation higher in energy than the staggered conformation.
- There is some steric repulsion b/w the H atoms in the eclipsed conformation that is reduced in staggered conformation.
Due to this:
i) Eclipsed - least stable, extraa energy (torsional strain) - more internal energy
ii) staggered - Most stable, less internal energy.
- Rotation

(I) (III) (V) Staggered (II) (IV) (VI) Eclipsed
- ENERGY

2) Butane
It has three carbon-carbon single bond and the molecules can rotate about each of them.
- Rotation

It has six structure
(I, III, V) - staggered conformers
(II, IV, VI) - Eclipsed conformers
- Conformer I - Most stable staggered, because in which two methyl group are as far (distance). Anti conformer (fully staggered).
- Conformer III & V - Gauche conformer (partial staggered)
- Conformer II & VI - Eclipsed (partial eclipsed)
- Conformer IV - Fully eclipsed (least stable)
- ENERGY DIAGRAM

stability fully staggered > Gauche > eclipsed > fully eclipsed.
3) Cyclohexane
- Acc. to Sachce & Mohr, cyclohexane shows 2 forms of structure
- Chair form staggered conformers (stable more).
- Boat form eclipsed conformers (less stable).
- Chair conformation is more stable than the boat conformation.

Energy diagram & Conformers
Conformations of cycloed cyclohexane are as follow
i) Chair form
ii) Half chair form
iii) Twist boat form
iv) Boat form
i) Chair Form
- There is no steric hindrance, so it has minimum energy and maximum stability.
- It is staggered conformers (most stable).
ii) Half chair form
- It has both angle strain and torsional strain, so less stable than chair form.
- Energy 46 kJ/mole.
iii) Twist boat form
More stable than boat conformation by about 5.4 kJ/mol, but less stable than chair conformation by 23.4 kJ/mol.
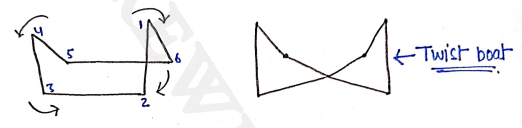
iv) Boat form
- Eclipsed conformers
- There are steric interaction b/w the non-bonding atom due to this, the boat conformation is less stable than chair conformation and has higher energy content.

Stereo isomerism in biphenyl compounds (Atropisomerism) and conditions for optical activity
- Atropisomerism : "Isolable stereoisomers resulting from restricted rotation about single bonds are called atropisomers and phenomena called as atropisomerism.
Atropisomerism Restricted Rotation around single bond $\rightarrow$ Stereoisomerism.
- There is restricted rotation about the bond between the two phenyl rings due to steric hindrance between the bulky ortho substituents.
- These phenyl rings lie in different planes which are perpendicular, thus the molecules becomes chiral and exhibits enantiomers.

Conditions for optical activity & for atropisomerism.
i) There should be any functional group/atom at ortho position of rings [substitution at ortho position with large size such as - Cl, Br, I, COOH, NO2, SO3H, CH3 etc-].
ii) Each ring must be resolvable, for that and .
Examples

Biphenyl atropisomers
Sterospecific and Stereoselective reaction
1) Stereospecific Reaction : [stereospecific synthesis]
- A reaction in which a particular stereoisomer react to give one specific stereoisomer of the product.
- In this reaction, stereoisomers gives different stereoisomere product. (stereospecificity). ea


Cis (Reactant) (Product) Trans
Trans (Reactant) (Product)
2) Stereoselective Reaction: [stereoselective synthesis]
- A reaction in which two or more stereoisomers formed possibly, but one stereoisomers is obtained more than the other.
Example :




