Pharmaceutical Organic Chemistry 3 - Unit 1
Syllabus
Stereo isomerism
Optical isomerism -
Optical activity, enantiomerism, diastereoisomerism, meso compounds
Elements of symmetry, chiral and achiral molecules
DL system of nomenclature of optical isomers, sequence rules, RS system of nomenclature of optical isomers
Reactions of chiral molecules
Racemic modification and resolution of racemic mixture. Asymmetric synthesis: partial and absolute
Scroll to Download
POC-3 UNIT-1
STEREOISOMERISM
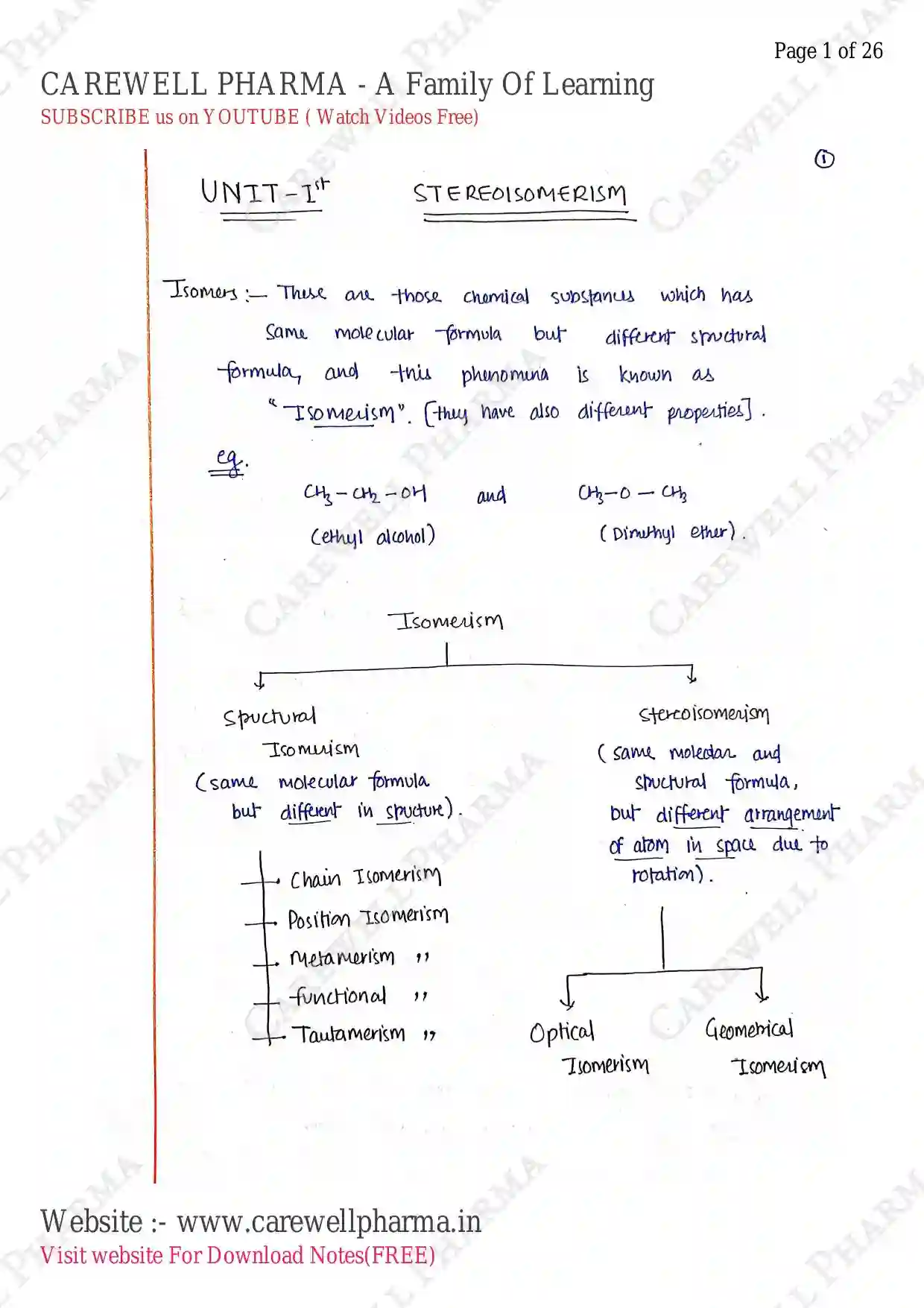
Isomers
These are those chemical substanus which has Same molecular formula but different spructural formula, and this phenomena is known as
"Isomerism". (they have also different properties].
(ethyl alcohol) and (Dimethyl ether).
Isomerism
- Spructural Isomerism (same molecular formula but different in spucture).
- Chain Isomerism
- Position Isomerism
- Metamerism
- functional Isomerism
- Tautamerism
- Stercoisomerism (same moledar and spuctural formula, but different arrangement of atom in space due to rotation).
- Optical Isomerism
- Geomerical Isomerism
Stereoisomerism
It is a form of isomerism in which molecules have the same molecular formula and same spructural formula but different in arrangement of atoms/groups in space.
eg : 1,2 dichloroethene
[Image description: Diagram showing Cis-1,2-dichloroethene and Trans-1,2-dichloroethene]
Optical isomerism
It isomerism is a type of stereoisomerism, in which Molecules have Same molecular, structural formula and also have Same physical properties, like melting point, boiling point, density ere-- But they differ in their behaviour towards Light.
- They also have same specific rotation of light but with opposite sign.
eg : Lactic Acid ($C_3H_6O_3$)
[Image description: Diagram of Dextro rotatory "D" or "+" lactic acid and Levo rotatory "L" or "-" lactic acid structures] (+) D-lactic acid L-lactic acid (-)
- Optical active compound which has power to rotate the plane polarised Light. (optical activity)
Optical Activity
The power of any substance to rotate the plane polatised light (PPL) from its axis to clockwise or anticlock wise direction is called "optical activity" And the substance is known as Optical active substance".
For Determination,

- Polarimeter, which check and measure optical activity.
- Firstly ordinary Light passes through Nicol prism, make it PPL (plane polarised Light), then pass this Light from polarimeter tube in which we filled a 1% solution which we have to Check optical activity.
- If the PPL goes unaffected or straight the solution/Compound is "optically inactive" but if the PPL rotate then compound (solution) is "optically active"
- Rotation is of two types
- If PPL rotates in clockwise direction, the solution is "Dextro rotatory" denote as "D" or "+".
- If PPL rotates in anticlockwise direction, the solution is "Levo rotatory" denote as "L" or "-".
Enantiomers
The optical isomers which are non-super imposable mirror image of each other are called "enantiomer"
eg : Lactic acid
[Image description: Mirror images of Lactic acid molecules that are non-superimposable]
- Enantiomers differ from one another in three dimensional Spatial arrangements.
- They have same melting point, density, solubility, colour and reactivity towards acids & bases.
- They have different specific rotation of plane polarised Light towards optical activity. A -> Dexno (+) Levo (-)
Diastereoisomerism
The stereoisomers that are non-mirror images of each other and also non-super imposable of each other, they have all Called "Diastereoisomers".
- Diastereoisomers have atleast two stereocentres.
- Diastereoisomers have different properties, such as melting point.

Elements of Symmery
Those compounds which can be divided into an equal parts from any plan called "symmetrical compounds"
I) Plane of Symmery : A plane which divides an object into two symmetrical halves is called "plane of symmury".

II) Centre of Symmeby : Also known as "point of symmetry".
- If all spaight lines that can be drawn through the centre of the molecule meet identical atoms at equal distance pom the centre.

Meso Compounds
A compound with two or more asymmunic Carbon atoms but also having a plan of Symmeby is called the "Meso compounds".
- Plane of symmetry, one half of the molecule is mirror image of other half. eg Tartaric acid

- These types of compound are optical inactive due to internal Compansation.
Chiral Molecules
Those molecules which cannot superimposable on its mirror image. (non-superimposable). these molecules are Optical active which rotati plane polarised Light.
Chiral means handedness.

- Chiral molecules also have a "chiral centre" or "Asymmetric carbon atom".
Chiral centre (Chiral carbon atom) - A carbon atom which attached with four differesht atoms/groups.
eg : Lactic acidChiral Carbon atom also known as asymmetric carban atom.
Achiral Moleuwles
- Those molecules which are superimposable on its mirror images.
- But these are Optical inactive molecules which can not rotati plane polarised light.
- eg : Dichlorofluoromethane

Representation of structure of Molecules
1) Wedge & Dash (flying wedge projection)
- These are the "3D representation" of Molecules.
- These are used for both chiral & achiral molecules.
- This protection is used for terahedral structure, in which two atoms/group are in plane and other two are in above and below the plane.

2) fischer Projection
- It is the "2D Representation" of molecules.
- In this method molewles are represented by cross line and the centre of lines is assumed as Carbon atom.

How to convert "Wedge-Dash projection" to "fischen projection"
- This conversion is important for the (D/L) and (R/S) configuration of molecules.
- In this, 3D stucture of molecules is convert into 2D structue.

- Those atoms/group which are on plane, represent all on vertical line in fischer projection.
- Then, watch wedge-dash structure from that side on which "above & below the plane" atoms/group attached.
- Then that atom/group which is in your left side, put it in left side and that atom/group which is in your right side, put in right side in "fischer projection".

NOMENCLATURE OF OPTICAL ISOMERS
1) D/L System :- (Relative Configuration)
The D/L system was developed by fischer and Rosanoff in around 1900.
- This method is used for optical active compounds.
- D(+) Dexborotatory: which has clockwise rotation OR Right hand side (R)
- L(-) levorotatory: which has anticlockwise rotation OR Left hand side (L) rotation.
- This D/L configuration is used for amino acid & Carbohydrates.
- In this Configuration, "Glyceraldehyde" is used as a reference compound.

Rules
I) Draw proper fischer projection formula of molecules.
II) Highur oxidised group must be on top. (if not then rearrange this).
III) Observe bottom most chiral (asymmubic) carbon, if it contain -OH/-NH2 at right then "D", if at left than "L".
- Longest chain is always on vertical line.
- If -OH and -NH2 both are present than D/L decided by -NH2 group.
Examples


R/S System of nomenclature of optical isomers
This is "absolute configuration", which is used for optical active Isomers compounds.
R Rectus [Right]
S Sinister [Left]
- This configuration was given by three scientist (European) R.S. Cahn, C.K. Ingold and V. Prelog. So, it is also known as "CIP system" [Cahn Ingold prelog].
SEQUENCE RULE (for RS configuration, It is necessary to know the sequence rule).
- firstly identify all chiral cenbe, then decide priority of all attached atoms/groups according to their "atomic number" i.e. higher atomic number with higher priority.
Eg : , their atm. number .
If the two or more atoms/group attached to the chiral centre are same, then give priority according to then "next atom/group"
The lowest priority atom/group must be on bottom or top of vertical line.
Some funcional group with their priority order
Rules for RS configuration
The atom/group attached with a chiral cenbe are assigned a number 1 to 4 according to "sequence rule".
Now, If the lowest priority group is on vertical line, then,
- Rotation of 1 2 3 is clockwise, configuration must be 'R' (Rectus).
- Rotation of 1 2 3 is anticlockwise, configuration must be 'S' (Sinister).
- But, If the lowest priority group is on horizontal line, then, (OPPOSITE)
- Rotation of 1 2 3 is clockwise, configuration is 'S'.
- Rotation of 1 2 3 is anticlockwise, configuration is 'R'.


Summary of ROTATION
- lowest priority group (LPG) should be on vertical line.
- Clockwise R
- Anticlockwise S
- But, if LPG (lowest priority group) is on horizontal line. Work Reverse (opposite).
- Clockwise S (change)
- Anticlockwise R (change)
Reactions of Chiral Molecules
Chiral Molecules:
- Those organic compound which has four different atom/group are attached with Carbon.
Racemic Mixture
The equimolar mixture of Dextro (D) / Levo (L) substance is called Racemic mixture.
Chiral molewles show three types of reactions-
I) Inversion
II) Racemisation
III) Retention
I) Inversion
These are those reactions in which chiral molecules change their nature after reaction, it convert
'R' [D] Dextro compounds 'S' [L] Levo compounds
'S' [L] Levo compounds 'R' [D] Dextro compoundseg : SN2 reaction show the inversion reaction.

II) Racemisation
In this reaction, optical active compound is converted into Optically inactive compound after the reaction.
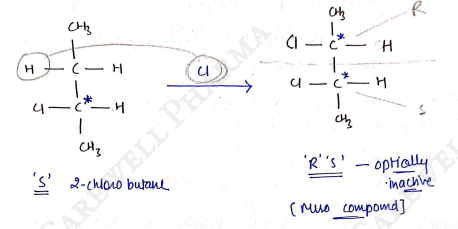
It this reaction, 'Cl' replace 'H' and form a meso compound, which is optically inactive.
III) Retention
- In this reaction chiral molecules/compound remain constant (same configuration) after reaction.

Racemic Modification and Resolution of racemic mixture
Racemic Modification
- Also known as Racemic mixture represented as (DL, RS, , dl).
- The equal amount of leavo (-) and dexro (+) forms Optically inactive mixture known as racemic modification. [-Equal amount of both enantiomers].

Resolution of Racemic mixture
- It is the process of separation of a racemic modification into its enantiomers constituents.
- Also in this process optically inactive compound is converted into optical active compounds.
Methods used for resolution
i) Mechanical method [ Crystallization mutnod]
ii) Chromatography
iii) Biological method
iv) Chemical Method
I) Mechanical method (Crystallization method)
- In this method, firstly crystallized the both enantiomers in racemic mixture.
- Now both have different shapes from each others. So, they are separated by magnifying lens and forceps.
- This is a mechanical separation (manual) given by Louis Pasteur in 1848.
- This method is applicable only to racemic conglomerates (If the molecules of the substance have greater affinity for Same enantiomers than for the opposite one).
- This method is economic but time consuming.
II) Chromatography
- Optically active substance can be resolved by chromatography.
- In this process, the racemic mixture is run through a column, which is filled with a optically active adsorbent (a chiral substance).
- The enantiomers will interacts differently with the chiral substance and elutes at different routes.
- In this optically active substance are adsorbed by optically active adsorbent Selectively on the basis of their affinity.
III) Biological method
- In this method, certain microorganism (bacteria, fungi, enzymes etc...) are allow to grow in Racemic mixture.
- They destroy one of the enantiomers at faster rati compare to other.
- Hence, the unchanged (other) enantiomer can be isolated by fractional crystallization or chromatography.
- It is developed by Pasteur in 1858.
- Drawback one enantiomer is sacrificed. and if mixture is toxic than they kill the microorganism.
- Eg. Penicillium used to remove D-ammonium tartarati from racemic mixture of ammonium tartarati.
IV) Chemical Method
- In this method a racemic mixture of enantiomers is converted into a mixture of diastereomers.
- The separation of enantiomers is difficult because of Same physical properties.
- So, enantiomer reacted with optially active compounds to form diastereomers.
- Then, these diastereoisomers can be separated due to difference in their physical properties and after separation they are reconverted to enantiomers by adding mineral acid ($H^+$).
- Eg : Acid-Base reaction often used for resolution of racemic alkaloids like quinine, morphine etc-.
Asymmetric Synthesis
In this method, Optically active compound is directly prepare from an Optically inactive compounds under the influence of some other optically active substance.

We can say that symmetric molecules (inactive) is directly converted into asymmetric molecules (active).
It is of two types
I) Partial Asymmetric Synthesis
II) Absolute Asymmetric synthesis
I) Partial Asymmetric synthesis
- In this asymmetric synthesis, Both (+) and (-) enantiomers are formed but one of these in is obtained in large amount.
- So, Both are not in equal amount and act as a Optically active compound and pass the plane polarised Light.
- Eg. Glucose etc-

II) Absolute Asymmetric synthesis
- In this synthesis, Racemic mixture (symmetric molecules) Is Completely convert into Optically active Compounds (Dextro OR Levo) any one.
- In this only one enantiomer can be formed completly.
- In this, any physical agent is used.