Pharmaceutical Microbiology - Unit 2
Syllabus
Identification of bacteria using staining techniques (simple, Gram’s &Acid fast staining) and biochemical tests (IMViC). Study of principle, procedure, merits, demerits and applications of physical, chemical gaseous,radiation and mechanical method of sterilization. Evaluation of the efficiency of sterilization methods. Equipments employed in large scale sterilization. Sterility indicators.
Scroll to Download
MICROBIOLOGY UNIT-2ND
Staining
It is a techniques, in which we identified the bacteria, by using different technique.
- There are different types of bacteria, which we can't see with naked eye, So we used microscope to see them, and use dye solution (as a indicator) for identification of bacteria.
Types of Staining
- Staining
- Simple staining
- Differential staining
- Gram technique
- Acid-fast staining
1) Simple Staining
In this staining, we observe the morphological characteristic [shape & size] of bacteria.
- In which we use single stain dye such as Crystal violet, Safranine, Methylene blue, Malachite green.
Principle
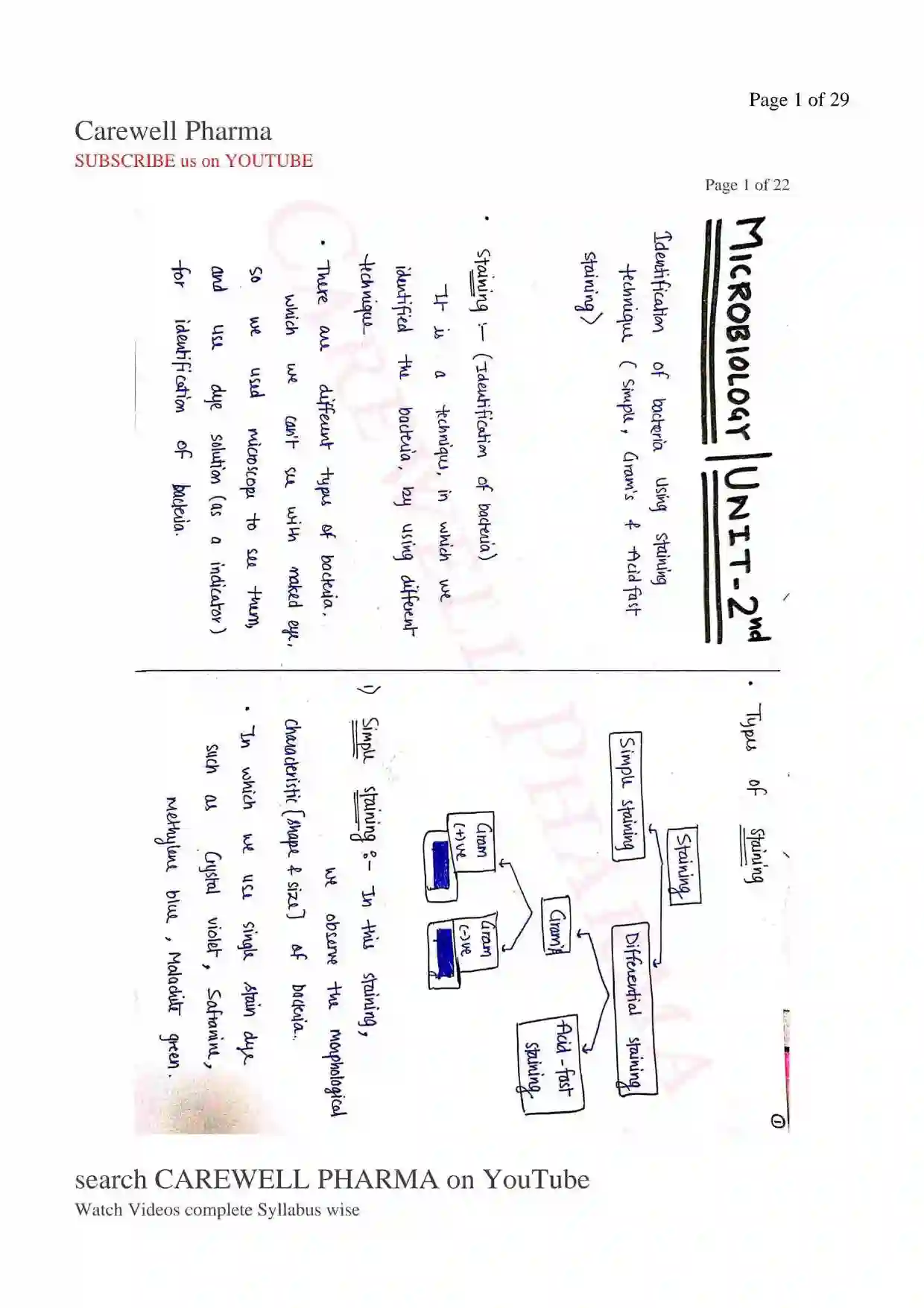
- Firstly develop the (-)ve charge on the surface of bacteria, so when we add (+) ve charge dye on bacteria, it will attached easily (-ve attract +ve and attached easily).
- When dye attached on the surface of bacteria, Bacteria will visible easily in also light background.
- Negative charge is develop on the surface of bacteria by releasing ion or adding ion.
Procedure
- Firstly take, Glass slide, cover slip, inoculation loop, culture media (bacteria present), Microscope.
- Then, wash all these with ethanol solution, after drying put under flamming for sterilization.
- Now, take inoculation loop, streak into culture media in which bacteria attached on loop, then streak inoculation loop on glass slide.
- Now, add some drop of any indicator on surface of slide (dye).
- Allow slide for drying, then wash the slide under tap water for remove excess stain.
- Now, wipe the below surface of slide with tissue paper.
- Then, Put the cover slip over the surface of slide, where bacteria stained, then placed slide on surface of microscope.
- Now see bacteria in microscope and obserbe the bacteria.
- The blue coloured spherical shape or Rod shaped bacteria is seen.
II) Gram's Staining
In this staining, we identified that, bacteria is gram (-)ve or gram (+)ve.
- This staining technique was developed by Han's christian gram in 1884.
- In this we use two or more dye at a time.
- On the basis of their structure, bacteria is classified into two category. Gram (+)ve and Gram (-)ve.
- Firstly we discuss some difference between them :-
| Gram (+)ve | Gram (-)ve |
|---|---|
| Peptidoglycon's multilayer present. | Peptidoglycon's single layer. |
| It's outer membrane absent. | It's outer membrane present. |
| Cell wall 20-30 nm thick & single layer. | Cell wall 8-12 nm thick & two layer. |
| After staining it gives purple/blue color. | After staining it gives Red/pink color. |
| eg. Streptococcus, bacillus etc- | eg. Eschercheia coli etc- |
Procedure
- Firstly take Glass slide, cover slip, inoculation loop, culture media (in which bacteria present) microscope (for see bacteria after staining).
- Now, wash all these with ethanol sol^n, then allow for drying, after drying put all these under flamming for complete sterilization.
- Now, take inoculation loop, streak into culture media in which bacteria attached on loop, then streak inoculation loop on glass slide.
- Now add some drops of crystal violet dye as a indicator and allow to dry.
- Now, add Gram iodine solution, which fix the crystal violet dye with cell wall.
- Now, wash the slide with ethanol and Acetone solution. [Gram (-)ve cell wall (8-12 nm) thin, so on washing crystal violet detached from Gram (-)ve bacteria, but Gram (+)ve cell wall thick (20-30 nm), so on washing crystal violet not detached and continuously attached on surface of bacteria].
- Now, add few drops of safranine dye on surface of slide and allow to dry.
- Then obserbe it on microscope.
Observation
- If purple/blue color obtained then bacteria is gram (+)ve, because crystal violet not detached on washing so Safranine not attached.
- If Red/pink color obtained then bacteria is gram (-)ve, because it detached crystal violet on washing with alcohol, so further when we apply safranine it attached with it and give red/pink color.
III) Acid Fast Staining
Also called as Ziehl-Neelsen staining.
- Discovered by scientist Paul Ehrlich in 1883.
- It is differential staining (use two or more dye).
- It is used for those microorganism, which is wax like and impermeable Cell wall (do not have cell wall).
- There microorganism does not identified by gram staining. so used acid fast staining, because it does not have cell wall.
Principle
- Used to identified that bacteria is Acid fast or Non-acid fast organism.
- When we add carbal fuschin dye as a indicator it react with inside material of bacteria in cytoplasm which is acidic and give red color to bacteria.
- On washing with alcohol Acid fast same red color but non-acid fast decolorized.
Procedure
- Firstly take glass slide, cover slip, inoculation loop, culture media (in which bacteria present) Microscope for see bacteria after staining).
- Now wash all these with ethanol sol^n, then allow for drying, after drying put all these under flamming for complete sterilization.
- Now take inoculation loop, streak into culture media in which bacteria attached on loop, then streak inoculation loop on glass slide and make smear on it.
- Now add few carbal fuschin dye (primary dye) on surface of bacteria and allow for dry.
- Then wash it with alcohol (Decolariser). (Carbal fuschin give red color to bacteria, on washing acid fast remain red, but non-acid fast decolorized and colorless.)
- Then further add some methylene blue (counter stain). (non-acid fast attached methylene blue & colored as blue, but acid fast not get it, because it is already colorized (red)).
- Again wash the slide with water, and then obserbe it by seeing it on microscope.
Observation
- If bacteria give Red/pink color it is Acid fast.
- If bacteria give blue color it is non-acid fast.
IMViC Test (Biochemical tests)
There are some kind of bacteria, which have same size and shape and which we can't identified through staining or microscope.
- So, identification of that type of bacteria are performed by chemically.
- We performed chemical test for this, and one of the chemical examination of any bacteria is performed by IMViC test.
- This is the biochemical series of test which is performed for differenciate into enterobacter.
Eg: enterobacter coli (E.coli), Enterobacter aerogenes (E. aerogenes) etc.
- I = Indole test
- M = Methyl Red (MR) test
- Vi = Voges-Proskauer (VP) test
- C = Citrate utilisation test.
I) Indole Production Test
Many bacterial species, which possess enzyme tryptophanase.
- Tryptophanase degrade amino acid tryptophan to indole, pyruvic acid and ammonia. Tryptophan Indole + Pyruvic acid + ammonia
- Inoculate the test microorganism into peptone water for check Indole production, incubating at for 48-96 hours (2-4 days).
- Then add 0.5 ml of kovac's reagent and mix gently.
- A red colour layer formed which indicates that positive reaction (production of indole).
- E. coli give Indole test positive, so it identified that E.coli present, if bacteria give Indole test positive.
II) Methyl Red (MR) Test
This test is performed to detect the production of acid during the fermentation of glucose.
- Due to production of acid, pH of the medium falls and it maintained below 4.5.
- Inoculate the test microorganism in glucose phosphate broth, then incubate at for 2 to 5 days.
- Then add few drops of 0.04% solution of Methyl red, mix well and obserb the result in the form of change in color.
- Red colour signifies positive (MR test). Yellow colour signifies negative (MR test).
- If test is positive, then E.coli bacteria. If negative, then E. aerogens bacteria.
III) Voges - Proskauer (VP test)
Many bacteria ferment carbohydrates of acetyl methyl carbinol (acetoin).
- In the presence of alkali and atmospheric oxygen, the small amount of acetyl methyl carbinol (acetoin) present in the medium is oxidised to diacetyl which react with the peptone of the broth to give a red color.
- Inoculate test microorganism in glucose phosphate broth, then incubate at for 48 hours (2 days).
- Add 1ml Potassium hydroxide (KOH) and 3ml of 5% solution of $\alpha$-naphthol in absolute alcohol.
- A positive reaction is indicated by the development of pink color in 2-3 minutes.
- VP test (E. aerogens give positive) & (E.coli give negative).
IV) Citrate Utilisation
In this test, check the use of citrate as a carbon source for growth.
- Koser's citrate medium has citrate as the source of carbon.
- Ability of microorganism to use citrate as the carbon source is indicated by the production of turbidity in the medium.
- So, finally if organism have ability to use citrate as carbon source, then it give positive test, if not then negative. (E. aerogen gives positive) (E. coli gives negative).
- Indole, MR, VP and citrate tests are done in rapid routine for the classification of Gram negative entric bacteria.
STERILIZATION
- It is a process in which remove or kill of all types of microorganism (bacteria, viruses, fungi.. etc) from the surface of all non-living or living things.
- It is an essential stage in the processing of any product use for parenteral administration, internal organs etc- and for cleaning of apparatus, glassware equipments etc.
Methods of sterilization
Physical Method - Dry heat sterilization. (Hot air oven), Moist heat sterilization. (Auto clave)
Chemical Gaseous Method - eg. formaldehyde, ethylene oxide etc--
Radiation Method - eg. , $\gamma$-rays, UV rays etc-
Mechanical Method - eg. filteration.
1. Physical Method
In this method, Heat apply on the surface, then proteins of bacteria are denatured and oxidised at high temp and it kill.
- It is easy & simple method.
- It is applicable only for thermostable substance, not for heat sensitive substance.
I) Dry Heat Sterilization
Heat is the most reliable and rapid method of sterilization. The killing effect of dry heat is due to protein denaturation, oxidative damage and the toxic effect of elevated levels of electrolytes.
Sunlight and Drying : This is natural method for sterilization of water in tanks, rivers and lakes. Sunlight possesses Ultraviolet rays (UV rays) which along with heat rays are responsible for appreciable germicidal activity. Drying is very unreliable method, without bacteria spores are unaffected by drying.
Red Heat : It is used to sterilize metallic objects by holding them on a flame till they are red hot. eg. inoculating wires, needles etc.
Flaming : Substance is passed over flame without allowing it to become red hot. eg glass slides, coverslip etc- (It destroy only vegetative microorganism)
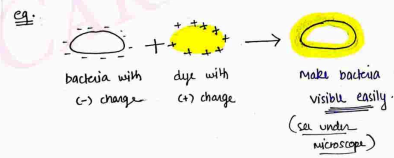
HOT AIR OVEN
This is the most widely used method of sterilization by dry heat.
Principle
In Hot air oven, maintain the high temp, so the protein of all bacteria are denatured and oxidised and the bacteria kills.
- It consist of double walled chamber of aluminium or stainless steel separated from the outer case by a thick layer of insulation made of fibreglass.
- Hollow flanged door carries asbestos jacket that provide tight seal. Heating is provided by electric heating elements.
Procedure
- Non-heatlabile substance put inside it.
- Normally the spores of bacteria as well as vegetative form of all microorganism are killed in two hours at a temp. of .
- The oven must be allowed to cool slowly for about two hour before the door is opened, since the glassware may get cracked by sudden cooling.
- Relation between temperature & time for hot air oven.
| Temperature ($^\circ C$) | Time (minutes) |
|---|---|
| 60 min | |
| 120 min | |
| 150 min | |
| 180 min |
Merits (Advantages)
- Simple and easy in use
- It does not release any harmful fumes.
Demerits (Disadvantages)
- Not suitable for surgical dressing, rubbers, plastic volatile and heat labile substance.
Applications (Uses)
- Used to sterilize Glassware forceps, Scalpels, scissors, spatula, etc-
- Also for some pharmaceutical substances such as glycerin, fixed oil, liquid paraffin, propylene glycol etc-
- Also used for some dusting powders such as kaolin, talc, zinc oxide, starch etc-
II) Moist Heat Sterilization
In this process, microorganism are killed by hot water or steam.
- It kills the microorganism by denaturation of enzyme and protein of microorganism.
- It is more effective than dry heat sterilization.
Moist heat sterilization is divided into three forms:
I) temperature below .
II) temperature at .
III) temperature above . (Auto clave)
I) Temp. Below
In this mostly heat-labile fluids may be sterilize by heating at temp. below .
Eg. Pasteurization of milk (Vaccines preparing from non-sporing bacteria may be inactivate in a water bath at for 1 hour).
II) Temp. at
Boiling at for 10 to 30 min kills all vegetative bacteria and some bacterial spores.
- An atmosphere of free steam is used to sterilize culture media which may decompose if subject to higher temperature.
- Not suitable for sterilization of instrument for surgical procedures eq Boiling etc.
III) Temp. above
AUTO CLAVE
Principle
- In which, saturated steam is produced under pressure.
- So, pressure increase in a closed vessels increase temperature proportionately,
- And, then high pressure is used to kill the microorganism.
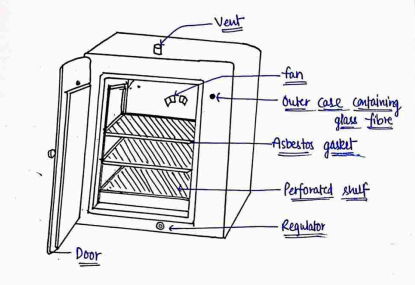
Construction
- It is consist of a verticle, cylinder of gun metal or stainless steel in a supporting frame or case.
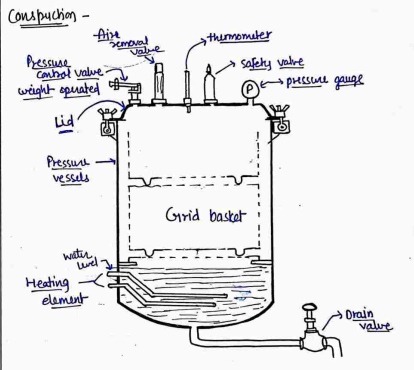
- The lid is fastened by screw clamps and rendered airtight by an asbestos gasket.
- The autoclave has on its lid or upper side a discharge tap for air and steam, a pressure gauge and a safety valve that can be set to blow off at any desired pressure.
Procedure
- The water is filled in autoclave and material is to be sterilization is load inside the autoclave.
- Now maintain the temperature and pressure for sterilization according to their need.
| Temp. ($^\circ C$) | Steam pressure (lbs/sq. inch) | Holding time (min.) |
|---|---|---|
| 115-118 | 10 | 30 |
| 121-124 | 15 | 15 |
| 126-129 | 20 | 10 |
| 135-138 | 30 | 3 |
- After put the material, lid is closed, discharge tap is opened and safety valve is adjusted to the required pressure.
- When air bubbles stop emitting from the discharge tap, it indicates all the air is removed from inside the autoclave.
- Then, close the discharge tap. Now, steam pressure rises inside and when it reaches the desired set level, safety valve opens and excess steam escapes.
- After the holding time (over), the heating is stopped and allowed to cool the autoclave (pressure gauge indicate it). Now, air is allow to remove from the autoclave by open discharge tap.
- Now, finally top lid is opened and the sterilized articles (material) are removed.
Merits (Advantages)
- It is very effective.
- It is quicker in heating up the exposed articles (material).
- It provide greater lethal action of Moist heat.
Demerits (Disadvantages)
- Not suitable for anhydrous material such as powders, oils, fats etc-
- Not suitable for heat labile substance, which cannot withstand heating at and above.
Applications
- It is used for sterilization bacteriological media, heat stable liquid, saline solution, heat resistant equipments and instruments, glasswares, filters, rubber products etc.
Chemical gaseous Method of Sterilization
When all types of microorganism killed by chemical gaseous. Is called chemical gaseous method of sterilization. eg. formaldehyde (HCHO), $\beta$-propiolactone (BPL), Ethylene oxide
I) Formaldehyde (HCHO)
- It is a bactericidal agent with poor penetrating power.
- It is generated by heating a concentrated solution of formaldehyde.
- It is an extremely active chemical, it combines readily with vital organic compound Nitrogen (N) such as proteins and (purines & pyrimidines) in DNA.
- So it destroy the pyrimidine or purines, which deactivate the DNA and bacteria killed.
- It is used for sterilization of enclosed area such as operation theatres, hospital rooms etc.
II) $\beta$-propiolactone (BPL)
- It is 25 times (approx.) more active as a disinfectant than formaldehyde gas.
- It is highly bactericidal and very active against viruses.
- It directly attack on the DNA of bacteria (microorganism). So, DNA damage, and due to DNA damage, bacteria killed. or the bacteria (microorganism) dies.
- It has a low penetration and show irritation so, it is not recommended for use in pharmaceutical applications.
III) Ethylene Oxide ($H_2C-CH_2$ bridged by O)
- It has high penetrating power.
- It attack on the DNA and RNA directly and react with it.
- It alkylating the amino, carboxyl, hydroxyl and sulphydryl group in the enzymes and protein molecules.
- Then it inactive the enzyme, protein of the microorganism, So the bacteria (microorganism) dies.
- Their are also some chemical liquids used for sterilization. eg Alcohol, Aldehydes etc...
- Alcohol : It dissolve with lipid in bacteria and denatured it, then bacteria dies.
- Aldehydes : It react with nucleic acid and protein of bacteria and killed it.
Advantages
- It is very light weight and not more expensive.
- It sterilized all places (even small places likes, holes etc-)
Disadvantages
- Very useful on used, it is harmful for humans.
- Give some side effect.
Applications
- Used for sterilization of medical and biological preparations.
- Used for sterilization of heat sensitive substances.
- Used in operation theatres, hospital rooms, microbiology laboratories.
Radiation Method of Sterilization
- When radiations (rays) are used for sterilization.
- This method also called cold sterilization, because very little amount of heat produced. So it is very helpful for sterilization of heat sensitive substances.
- Based on their wavelength and penetrating power, divided into two parts-
I) Ionizing Radiations
- It have high energy and it ionize the target molecules.
- When energy (rays) passes on bacteria it change the base pair of DNA, due to this mutation occurs in DNA.
- Due to mutation, Bacteria's DNA structure and function changes and DNA damaged.
- After damaged DNA, bacteria dies.
- The sources of energy are used in this method:- X-rays, gamma rays and Cathode rays.
II) Non-ionizing Radiations
- have low energy and it is used for hospital operating rooms, in aseptic filling room in pharmaceutical industry.
- When rays (energy) passes through bacteria. nucleic acid (DNA) of the cell absorb it.
- It does the great damage, it induce the production of abnormal nucleotides such as thymine dimers.
- These interfere the process of DNA replication, which stop the formation or impare formation of adenine etc.
- Due to this, DNA (Nucleic acid) damage, and the bacteria (microorganism) dies.
- The sources of energy (rays) are used in this: UV rays (widely used), IR rays.
Advantages
- Helpful for sterilization of heat sensitive materials.
- It is less time consuming.
- Very effective for reduction of air-borne contamination.
- Probably all bacteria kills.
Disadvantages
- It have very expensive.
- It may causes side effects.
- In non-ionizing, penetrating power being negligible (very less) so its effectiveness is limited to exposed surface only.
Applications
- Used in food and dairy industries for treatment of contaminated surfaces.
- Used for sterilizing biological fluids such as blood plasma and vaccines.
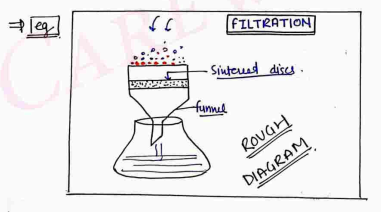
Mechanical Method of Sterilization
- Based on filteration.
- It is simple method in which solution (material) passed through bacteria-proof filter unit, then bacteria is removed from solution.
- The process of sterilization by filteration consist of following stages:-
- Passage of the solution through a previously sterilized bacteria-proof filter.
- Aseptic transfer of the filtrate to sterile containers which are then sealed aseptically.
- Now, bacteria is remove and stay on filter, which further pure solution (sterile) get.
- Testing of sample for sterility.
Different types of filter used in it
I) Seitz filter (Asbestos filter)
II) Sintered glass filter
III) filter candles (ceramic filter)
IV) Membrane filter (millipore/ultra filter).
Advantages
- Helpful for sterilization of heat-labile solution.
- Simple and easy in use.
- Used for large volume solution, eye drops, antibiotic solutions etc.
Disadvantages
- Not 100% reliable, it required test for sterility.
- Many medicinal preparation adsorb on the surface of filter.
Applications
- Used in purification for many pharmaceutical products.
- Used for sterilization of antibiotic solution, serum, Carbohydrates etc.
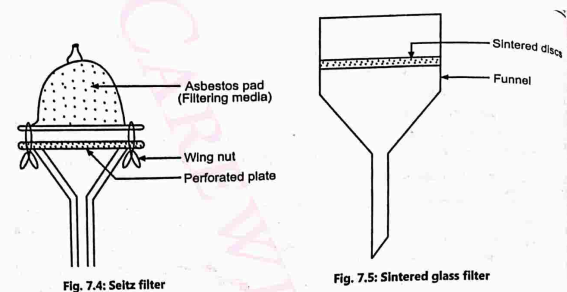
(i) Asbestos filter (Seitz filter)
They are disposable, single-use discs made up of asbestos (magnesium trisilicate). It is supported on a perforated metal disc within a metal funnel (Fig. 7.4). It is then fitted on to a sterile flask through a silicone rubber bung. The fluid to be sterilized is put into the funnel and flask is connected to the exhaust pump. After completion of filtration, the filter is discarded and the entire unit sterilized. The pore size of filters range from 0.01 to 5 microns.
(ii) Sintered Glass Filters (fritted glass filter/morton filters)
Borosilicate glass is finely powdered in a ball mill and packed into disc moulds and heated until suitable adhesion takes place between the granules. The sintered discs are finally fused into funnels of a suitable size and shape (Fig. 7.5). Sintered glass filters are available in several different porosities but for filtration sterilization a number or grade 5 or 5 on 3 must be used. They have a low adsorptive property and can be cleaned easily. They are brittle and expensive and have a small area of filtration.
(iii) Filter Candles (ceramic/Berkefield filter)
These are manufactured in different grades of porosity and have been used widely for purification of water for industrial and drinking purposes. They are made of either porous porcelain or kieselguhr. They are usually encountered as cylindrical candles with comparatively thick walls (Fig. 7.6). These are depth filters with cellular walls and are available in various sizes.
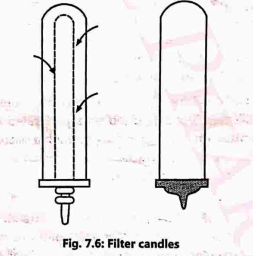
The filter is fixed to the filter assembly and placed in a mantle. The liquid to be filtered is poured into the mantle where vacuum forces it through the filter. After filteration, filter candle is removed from the assembly and filterate is transferred to a sterile container.
These filters are inexpensive and available in different sizes. They are easily clogged and blocked and require high pressure for filtration.
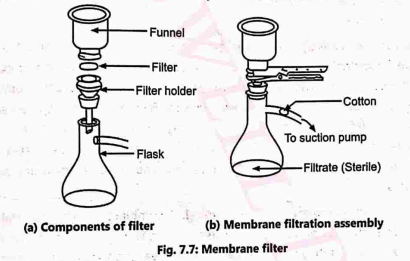
(iv) Membrane Filter (millipore/ultra filter):
These are made up of various types of cellulose and cellulose esters. They are thick and contain millions of microscopic pores ranging from 0.01 to in diameter. The pore sizes most often used for sterilization are (Millipore grade, HA) or (Millipore grade, GS), particularly for very small bacterial contaminants. They are sterilized by autoclaving, in the holder or packed between thick filter pads to prevent curling. They are also available at ready sterilised form (by ethylene oxide or ionizing radiation). Membrane filters are supported on a rigid base of perforated metal, plastic or coarse sintered glass (Fig. 7.7). The HA grade filters are approximately 65 ml/min./sq.cm (GS-22 ml/min/sq.cm) with a differential pressure of 70 cm mercury across the membrane.
Advantages of Membrane Filters are as Follows
- All microorganisms are separated by process of sieving.
- Membranes have a high and uniform porosity permitting a rapid rate of filtration.
- Membranes are disposable. Hence, there is no cross contamination between filtered products.
- Adsorption is very less.
Disadvantages of Membrane Filters are as Follows:
- Prefilter is used before the membrane filter to avoid clogging and breaking.
- They have less chemical resistance to certain organic solvents such as chloroform, ketones and esters.
Membrane filters are routinely used in water purification and analysis, sterilization, sterility testing and for the preparation of solutions for parenteral use. They are also been used for the identification and enumeration of microorganisms from water samples and other materials.
Evaluation of the efficiency of sterilization method
To check the ability of sterilization process for killing bacteria (microorganism), and finally ensure that all microorganism are killed.
It can be determined by three value
I) D-Value
II) Z-Value
III) f-Value
I) D-Value (Decimal reduction time)
Time in minutes at any constant (defined) temperature to destroy 90% viable microorganism is called D-Value.
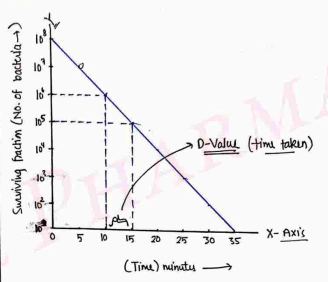
- If the value of D decrease (less), then efficiency of sterilization very effective, because D value is, time required for destroy microorganism, so if in less time (D value) more bacteria kill, then sterilization very efficient.
II) Z-Value
It is the number of temp. change required to reduce one point in D-Value.
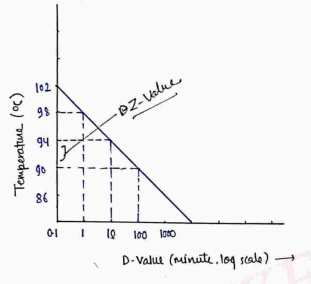
- Z value is change in temperature.
- So, On increasing temperature more bacteria will kill and the D-value is decrease.
- Z-value is reciprocal to D-value.
- And, less D-value means efficiency of sterilization is increase.
III) f-Value
The time required in heating method of sterilization to kill the population of bacterial spores in minutes is called f-value.
- The f-value can be used to calculate the probable number of survivors remaining in a load as:-
Where,
= D-value at of the organism
= initial population number/unit volume
= final population number/unit volume
Sterility Indicators
It is essential that strict controls are carried out on products to be labelled 'sterile'. Such controls must then ensure the absence of viable microorganisms from these products.
There are basically two types of controls
- Controls on the process of sterilization i.e. sterilization monitors or sterilization indicators.
- Sterility testing of the products.
Monitoring of the sterilization process can be achieved by the use of physical, chemical or biological indicators of the sterilization performance.
1. Physical indicators
(i) Moist heat
A master process record (MPR) is prepared as part of the validation procedure for a particular autoclave and for each specified product and load configuration. This may then be used as a reference for the process record obtained from a single thermocouple placed in a strategic part of each load (batch process record, BPR). The MPR should be checked at annual intervals and whenever significant changes occur in the BPR when compared with the MPR. Microprocessor controlled sterilization cycles are now a part of modern autoclaves. Pressure is measured by pressure gauges or through pressure transducers.
(ii) Dry Heat
In dry heat sterilization processes, a temperature record chart is made of each sterilization cycle and is compared against a master temperature record.
(iii) Radio Sterilization
A plastic dosimeter gives an accurate measure of the radiation dose absorbed and is considered to be the best technique currently available for the radio sterilisation process.
(iv) Gaseous Methods
For gaseous sterilization procedures, elevated temperatures are monitored for each sterilization cycle by temperature probes and routine leak tests are performed to ensure gas-tight seals. Gas concentration is measured independently of pressure rise, often by reference to the weight of gas used. Pressure and humidity measurements are recorded.
(v) Filtration
Bubble point pressure test is a technique employed for determining the pore size of filters and may also be used to check the integrity of certain types of filter devices immediately after use. The principle of the test is that the filter is soaked in an appropriate fluid and pressure is applied to the filter. The pressure difference when the first bubble of air breaks away from the filter is equivalent to the maximum pore size. When the air pressure is further increased slowly, there is general eruption of bubbles over the entire surface. The pressure difference is equivalent to the mean pore size.
2. Chemical indicators
Chemical monitoring of a sterilization process is based on the ability of heat, steam, sterilant gases and ionizing radiation to alter the chemical or physical characteristics of a variety of chemical substances.
(i) Browne's Tubes
The most commonly used chemical indicators for heat processes are Browne's tubes. These are small sealed tubes containing a reaction mixture and an indicator. Exposure to high temperature completes the reaction producing a change in the colour of the indicator (Table 7.5). All four types change from red through yellow brown to green, the latter colour only being achieved after a specified time at the given temperature.
| Browne's tube | Method of sterilization | Temperature ($^\circ C$) | Colour of indicator |
|---|---|---|---|
| Type I | Moist heat | 126 | Black spot |
| Type II | High vacuum moist heat | 130 or more | Yellow spot |
| Type III | Dry heat | 160 | Green spot |
| Type IV | Dry heat infra-red conveyor oven | 180 | Blue spot |
(ii) Witness Tubes
Witness tubes consist of single crystalline substances of known melting point contained in glass tubes e.g. sulphur ($115^\circ C$), succinic anhydride ($120^\circ C$), benzoic acid ($121^\circ C$) etc. A dye may be included to show more clearly that the crystals have melted. Such a device only indicates that a certain temperature has been reached. Exposure time can be calculated by putting the crystals in one end of an 'hour-glass' tube, the volume of the crystals and the diameter of the constriction of the tube being adjusted so that the time for transfer of the melt is the same as that required for the sterilisation at the required temperature.
(iii) Heat-Sensitive Tape
Heat-sensitive tape is used quantitatively in the Bowie-Dick test. This is a test to determine that all air has been removed from dressings and that subsequent steam penetration has been even and rapid. The tape is placed suitably wrapped at the centre of a test pack. All the bars on the tape should change colour to demonstrate full penetration of the steam.
(iv) Royce Sachet
The Royce sachet is a chemical indicator for ethylene oxide sterilization. This consists of a polythene sachet containing magnesium chloride, HCl and a bromophenol blue indicator. A given concentration-time exposure to ethylene oxide results in the formation of ethylene chlorohydrin and a colour change from yellow to purple.
(v) Chemical Dosimeters
Chemical dosimeters give an accurate measure of the radiation dose absorbed and are considered to be the best technique currently available for controlling radiation sterilization. Qualitative indicators made of radiosensitive chemicals impregnated in plastic are also available. The indicator changes from yellow to red during irradiation.
3. Biological indicators
Biological indicators consist of a suitable organism deposited on a carrier and are distributed throughout the sterilizer load. At the end of the sterilization process, the units are recovered and cultured to determine the presence or absence of survivors. The biological indicator measures sterilization processes directly and is able to integrate all sterilization parameters. The selected organism should possess high and reproducible resistance to the sterilizing agent, should be genetically stable, readily characterizable and non-pathogenic. The viability of the organisms, the storage conditions before use and the incubation and culture conditions after sterilization must be standardized for the results. The organisms used as biological indicators are usually resistant bacterial spores.
Biological indicators for monitoring sterilization processes
| Sterilization process | Species | D-value |
|---|---|---|
| Autoclave at | Bacillus stearothermophilus | 1.5 min |
| Dry heat at | Clostridium sporogenes | 0.8 min |
| Ethylene oxide at (Temperature 60% relative humidity) | Bacillus subtilis var. niger | 5-10 min |
| Ionizing radiation | Bacillus pumilus | 2.5 min |
| Membrane filter ($0.45~\mu m$ pore size) | Serratia marcescens | 3 kGy (0.3 M rad) |
| Membrane filter ($0.22~\mu m$ pore size) | Pseudomonas diminuta |