Pharmaceutical Inorganic Chemistry - Unit 5
Syllabus
Radiopharmaceuticals: Radio activity, Measurement of radioactivity, Properties of α, β, γ radiations, Half life, radio isotopes and study of radio isotopes - Sodium iodide I131, Storage conditions, precautions & pharmaceutical application of radioactive substances.
Scroll to Download
RADIOPHARMACEUTICALS
UNIT - 5TH
SYLLABUS :
Radiopharmaceuticals: Radio activity, Measurement of radioactivity, properties of , , radiations, Half life, radio Isotopes and study of radio isotopes sodium Iodide , storage conditions, preparations & pharmaceutical application of radio active substances.
Radiopharmaceuticals
These are radioactive substances which are used therapeutically for treatment and diagnosis.
- these substances get distributed in the body and emit radiations, which used to detect injury, abnormal cells growth etc..
- when these rays passes, they penetrate the body tissue, muscle but not bones which gives x-ray images, helps to identified injury & fractures etc.
Radioactivity
It is phenomena in which radioactive substances or radioisotopes [which are unstable] undergoes decomposition or decay by emission of radiation/rays to get stable.
- radiations are emitted from the nuclei of element.
- there are various types of radiations, but the most common are alpha, Beta and Gamma.
- A branch of Pharmacy which deals with radioactive substances are known as Radiopharmacy.
RADIATION
Radioactive substances emitted radiations which is travel through a medium/space and absorbed by another medium.
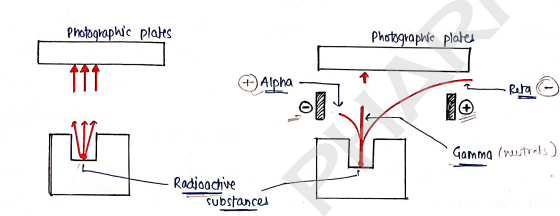
1. Alpha rays ($\alpha$-rays]:
- These are positively charged particles and heavy.
- They have the least penetrating power.
- These particles are equivalent to the nuclei of helium atoms (i.e. $He^{2+}$).
- They are highly energetic particles and have energy upto . $$^{226}Ra \rightarrow ^{222}Rn + ^{4}_{2}He (\alpha)$$
- less/not useful in p'ceutical formulation.
2. Beta rays/radiation [$\beta$-rays]:
- They are mostly negative charged particles (negatron or electron), but rarely may also be positively charged. [positron]
- They have great penetrating power than that of the $\alpha$-rays.
- Their mass is equal to an electron [approx. of unit mass] $$^{14}{6}C \rightarrow ^{14}{7}N + \beta + e^{-}$$
- The maximum energy exhibited by beta particles (E_{max}$) is $1.5 MeV and mean energy (\bar{E}$) is $0.6 MeV.
3. Gamma radiation [$\gamma$-rays]
- Gamma particles are similar to X-radiation.
- It is an electromagnetic radiation, therefore does not have any mass or charge. It is neutral.
- They have more penetrating power than alpha and beta rays, and travel with the speed of light.
- Gamma particles are high energy particles, typically .
APPLICATIONS OF RADIATIONS
- Radiations are used for diagnostic purposes. x-rays.
- used in sterilization as they destroy bacteria & other microbes.
- various radioactive isotopes are used to supply power to satellites and provide electricity for space laboratories.
- they are utilised for estimating the degree of air pollution.
UNITS OF RADIOACTIVITY
It is the no. of disintegrations per second.
- Curie ($Ci$) : *
- It is the conventional unit.
- One curie is defined as activity of material which has disintegration per second.
- Becquerel ($Bq$): It is SI unit. $1 Bq = 2.703 \times 10^{-11} Ci$ [1 disintegration per second].
- Roentgen ($R$): International system of units etc.. $1 R = 2.58 \times 10^{-4} C kg^{-1}$ [ = Coulomb]
Decay Constant
It is the actual amount of emission of radiation/ray per unit time.
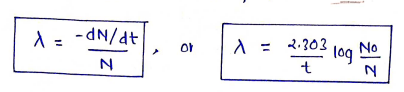
where, radioactive decay or disintegration constant. quantity, time.
Half-life
It is the time period in which a substance or radionuclide is reduced by or half of its initial amount.
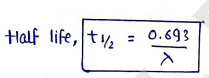
where, is disintegration constant in unit of .
- Iodine-131 8 days
- Polonium-212 seconds
- Uranium-238 years etc..
MEASUREMENT OF RADIOACTIVITY
The radioactivity of alpha, beta and gamma can be measured by various techniques :-
- Ionisation chamber
- Geiger-Muller counter
- Scintillation counter
These techniques mainly work by detection and counting of radiations.
1. Ionisation Chamber
- It simply measure the radiation strength by measuring the strength of current.
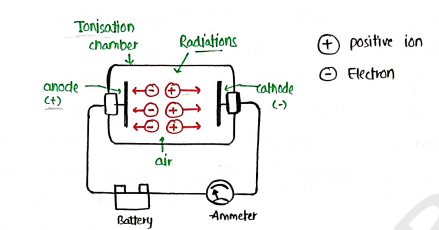
- Ionisation chamber is filled with two metallic plates, separated by air.
- on passing the radiation through the chamber, the atoms of air molecules are knocked off and the charged formed.
- The electrons (-ve) move towards the anode while positive ions towards the cathode, which formed the current measured in ammeter.
- The total amount of charge passing b/w the plates in a given time is measured by dosimeter, present in ionisation chamber.
2. Geiger-Muller counter
The rate of emission of or $\beta$-particles can be detected and measured by this geiger-muller counter.
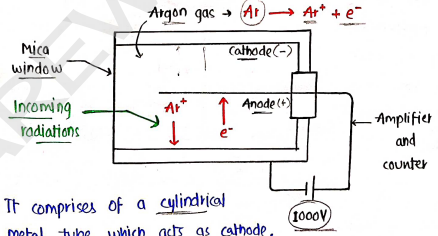
- It comprises of a cylindrical metal tube which acts as cathode, and for anode a wire is placed carefully.
- The metal tube has very low pressure of around and filled with argon gas.
- A potential difference of 1000 volts is maintained across electrodes.
- Radiations enters through mica window and argon gas ionised and move towards cathode while electro move towards anode.
- The circuit is completed in very short time (in microseconds), after which an electric pulse is generated which is recorded in the automatic counter.
- The intensity of radioactivity is measured by the pulse generated per minute.
3. Scintillation counter
It works on detecting a flash or scintillation of light, which is produced when a charged particle, or X-rays & gamma rays, strikes on either fluorescent screen.
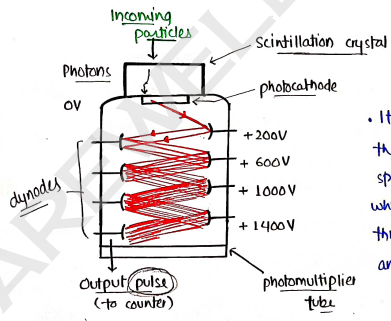
- It is based on that the radiation enter is strikes on photocathode, which then multiply through dynodes [all anodes] and collected at output.
- then this output pulse is measured, which is directly proportional to the intensity of radiations.
RADIOISOTOPES
A radioisotope is an atom with an unstable nucleus i.e. an unstable combination of neutrons and protons. Now, these atoms emitted radiations/rays to make itself stable.
- As a isotopes they have same no. of proton but varies neutrons.
Types :
Stable radioisotopes: these are stable and do not emit radiations.
, , (protium), (deuterium) etc..Radioactive radioisotopes: they are either naturally or artificially produced unstable isotopes. so, they emit radiations to loose energy. phenomena is known as radioactivity.
Study of Radioisotopes :
- Carbon element : It has three isotopes

Iodine element : It has two isotopes
- Iodine-127 53 protons, 74 neutrons stable isotope
- Iodine-131 (^{131}_{53}I$) $\rightarrow 53 protons, 78 neutrons, unstable isotope.
Phosphorus element : It has two isotopes:-
- phosphorus-31 (^{31}_{15}P$) $\rightarrow stable
- phosphorus-32/32P (^{32}_{15}P$) $\rightarrow Unstable
PRECAUTIONS IN HANDLING & STORAGE OF RADIOP'CEUTICALS
- Great care has to be taken in handling and storage of radioactive material for protecting people and personnel handling it.
- One should not touch radioactive emitter with hand but it should be handled by means of forceps or suitable instruments.
- While handling liquid radioactive materials, rubber gloves should be used.
- The radioactive material should not contaminate the working area.
- Smoking, eating and drinking activities should not be done in the laboratories where the radioactive material are handled.
- Sufficient protecting clothing and shielding devices have to be used while handling the materials.
- Radioactive substances are suggested to be used within a short period time.
- The disposal of radioactive material is done with great care.
STORAGE :
- The radiopharmaceuticals are stored in airtight containers in a shielded place, and in suitable labelled containers.
- Stored place should be monitored as per National/International regulations for radioactive substances.
- The containers of radiopharmaceuticals may become dark due to irradiation. & optimum storage condition should be maintained.
- All the requirements are prescribed by the department of atomic energy [DAE] for the establishment of a radio-isotope facility in a hospital or pharmacy.
PRODUCTION OF ISOTOPES/RADIOISOTOPES
a) Reactor nuclear irradiation : A reactor is having a moderator, which slow down the fast neutrons to thermal energies.
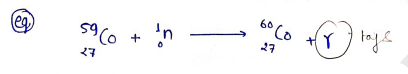
b) Cyclotrons reactors:- Radionuclides stable by positron emission or nuclei, therefore resultants.
Therapeutic Applications :
- These radiations have destructive features, so destroy abnormally multiplied cells and further inhibit the formation of new cells and tissue.
- Used in the treatment of disorders like cancers which involves cellular malfunction.
- (e.g. Iodine-131 [sodium iodide] used for thyroid scanning. diagnosis of various organs imaging. e.g. for myocardial)
3. Sterilization :
- Radiations are used to sterilise the pharmaceutical and surgical instruments in hospitals.
- Cerium-137 may be used for sterilising surgical instruments.
4. Research :
- Excellent biological and medicinal studies have been carried out with radio-active isotopes as tracers.
- e.g. and are most commonly used radio-nuclides for this.
SODIUM IODIDE
It is a radiopharmaceutical substance used in the treatment of malignant thyroid. The ionising radiations of Iodine are absorbed by the thyroid tissue and damage that tissue.
- The sodium iodide emits about of \beta$-radiations and the remaining $10\% is the $\gamma$-radiation.
Mechanism : The iodide enters into thyroid through the sodium/iodide symporter and accumulates there. Here it oxidises into iodine and emits radiations. Now, these $\beta$-radiations destroy the thyroid tissue.
Properties :
- It is a colorless solution having a pH between .
- Half-Life of sodium iodide is days.
- It emits $\beta$- and $\gamma$-radiations.
Uses :
- It is used in the treatment of hyperthyroidism and some cases of thyroid malignancy.
