Pharmaceutical Inorganic Chemistry - Unit 2
Syllabus
Acids, Bases and Buffers: Buffer equations and buffer capacity in general, buffers in pharmaceutical systems, preparation, stability, buffered isotonic solutions, measurements of tonicity, calculations and methods of adjusting isotonicity.
Major extra and intracellular electrolytes: Functions of major physiological ions, Electrolytes used in the replacement therapy: Sodium chloride\*, Potassium chloride, Calcium gluconate* and Oral Rehydration Salt (ORS), Physiological acid base balance.
Dental products: Dentifrices, role of fluoride in the treatment of dental caries, Desensitizing agents, Calcium carbonate, Sodium fluoride, and Zinc eugenol cement.
Scroll to Download
ACID BASES BUFFERS
Chapter-1
Unit-2
SYLLABUS : Introduction, theories of Acid and Bases, Importance of acids and bases. Buffers - Introduction, Buffer Capacity, Buffer Equation (Handerson - Hasselbalch Equation), characteristics of buffer solution, Types of Buffer solution, Buffers in pharmaceutical systems, Buffered Isotonic Solutions, Pharmaceutical Importance of Buffers.
ACIDS AND BASES
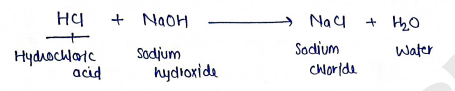
These are popular chemicals which interact with each other in the formation of salt and water. $$HCl + NaOH \rightarrow NaCl$$
Acids:
- These are those substances which can be identified by their sour taste.
- An acid is a molecules which can donate an ion (proton ions).
- Acids are known to turn blue litmus red.

- eg : , , , Vinegar, Lemon Juice, Vitamin C etc..
Bases :
- These are those substances which can be identified by their bitter taste, slippery texture.
- A base is a molecules which can accept a hydrogen ion ($H^+$) or release ions.
- Bases are known to turn red litmus blue.

- eg : , , Baking Soda, Soaps, Ammonia ($NH_3$).
THEORIES OF ACID AND BASES
These are those theories which are basically used to define and explain the concept of Acids and Bases.
It is of three types :
- Arrhenius theory
- Bronsted-Lowry theory
- Lewis acid-base theory
1. Arrhenius theory
It is most commonly used concept for acids and bases, and it is developed by Savante Arrhenius in 1884.
According to this theory :
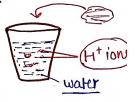
- Any substance which produces/gives ions when dissolved in aqueous solution (water) are known as Acids.
- Example:
- Any substance which gives ions when dissolved in aqueous solution (water) are known as Bases.
- Examples etc..
It is the first scientific theory and the simplest theory used in case of aqueous solution.
Limitations :
- This theory explain the acidic and basic solution only in aqueous medium and fails to explain in non-aqueous medium.
- There are many acids or bases which does not contain ions but they are acidic/Basic in nature. eg (Bases), (Acids).
2. Bronsted-Lowry theory
J.N. Bronsted and J.M. Lowry in 1923 gave a broader concept of acids and bases. According to this theory:
- Acids: These are those substances which are capable to donate the 'proton' ($H^+$ ion) are known as Acids (Proton Donor).
- Base: These are those who accepts the proton ($H^+$) are known as Base (Proton Acceptor).
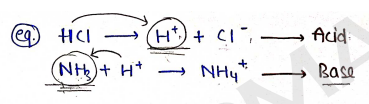
Conjugate Acid-Base Pairs : In Acid-Base reaction, when acid react with base it produces a new base by donating its proton ($H^+$) and that base is known as conjugate base.
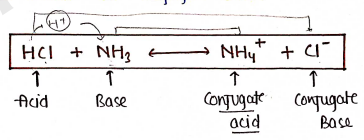
- The substance which accept protons are known as conjugate acid.
Limitation :
It fails to explain about those molecules which does not contain ions.
eg : etc..
3. Lewis theory of Acids and Bases
This theory was given by G.N. Lewis in early 1930s.
According to this theory:
- Acids : Those substances which accepts the lone pair of electron are known as acids, i.e. Electron-pair acceptor (Lewis acid).
eg : etc...
- Base : Those substances which have lone pair and donate the lone pair of electrons are known as Base or lewis Base i.e. Electron pair donor.
eg : (ethylene) etc...
- A Lewis acid accepts an electron pair from a lewis base, forming a coordinate covalent bond which form Lewis adduct complex.
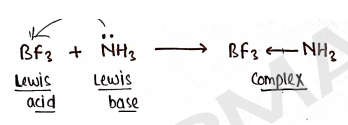
Limitation : It fails to explain the acid-base reaction that do not involve the formation of a coordinate covalent bond.
Importance of Acids and Bases
- Used in the preperation of suitable salts.
- Various acids and bases are used during acid-base titrations.
- Various acids and bases are found in human body and used to maintain pH of GIT, Urine, blood. HCl for digestion.
- Used in daily households. Vinegar, Citric acid (lemon) and Sulphuric acid, Soaps etc..
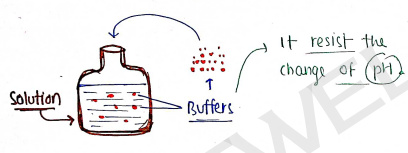
BUFFERS OR BUFFER SOLUTIONS
These are those solutions which resist the change of pH of solution, when a small amount of either an acid ($H^+$ ions) or a base ($OH^-$ ions) is added.
- Eg : Ammonium acetate solution.
Characteristics of buffer Solution:
- It has reserve acidity or alkalinity, thus a definite pH.
- It does not change pH, even if stored for long duration.
- There is no alteration in its pH on dilution.
TYPES :
It is of two types:
- Acidic Buffers
- Those solutions which are added in the acidic solutions.
- It is made up by mixing a weak acid and its salts with a strong base.
- eg. (Acetic acid) (Sodium acetate)
- Basic Buffers
- Those buffers which are added in the basic solutions.
- It is made up by mixing a weak base and its salts with a strong acid.
- eg. NH_4OH + NH_4Cl$ (Ammonium hydroxide) (Ammonium chloride)
Buffer Capacity
The amount of acid or base that must be added to the buffer to produce a unit change of pH. It helps to know the effectiveness of a buffer on a quantitative basis.
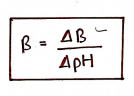
where,
- = Buffer Capacity
- = Amount of acid/base
- = pH changes
BUFFER EQUATION
Also known as Handerson - Hasselbalch Equation.
It is used to calculate the pH of a buffer solution and the changes in pH which occurs during addition of an acid or base.
For Acidic buffers [weak acid and its salts] :
The pH of acidic buffer can be calculated from the dissociation constant ($Ka$) of the weak acid and its salt.
- let's take (weak acid) and (salt).

- Taking on both sides,

BUFFERS IN PHARMACEUTICAL SYSTEMS
The buffers play an important role in pharmaceutical preparation to ensure pH condition for the medicines.
- to maintain solubility of pharmaceuticals by adjusted its pH.
- to maintain stability of pharmaceuticals.
- to preserve pH of pharmaceuticals.
Eg : Phosphate buffer, Borate buffer
BUFFERED ISOTONIC SOLUTIONS
These are those solution which have same osmotic pressure as that of body fluids. These solution are meant for application of body.
- Blood = 0.9% w/v NaCl solution.
Tonicity : It is defined as it is the amount/concentration of solute in any solution.
- eg. 0.9% w/v NaCl solution i.e. 1gm NaCl is dissolved in 1000ml of water.
It is of three types :
Isotonic solution : A buffer solution that have same osmotic pressure as body fluids i.e. 0.9% w/v NaCl solution.
Hypotonic solution : A buffer solution that have less solute concentration (osmotic pressure) than 0.9% NaCl solution. (< 0.9% NaCl)
Hypertonic solution : A buffer solution that have high solute concentration (osmotic pressure) than 0.9% NaCl solution. (> 0.9% NaCl)
- The concept of isotonicity is used during preparation of medicament/I.V. fluids for body. Because, if the tonicity of these medicament is different than blood/body fluids, then they cause irritation or maybe serious damage.
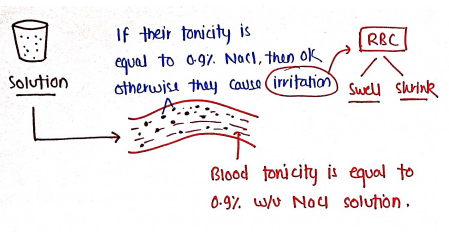
- So, Isotonic solution are those solution which contain 0.9% w/v NaCl solution.
Method to determine Isotonicity
- Cryoscopic method (Colligative methods)
- Hemolytic Method
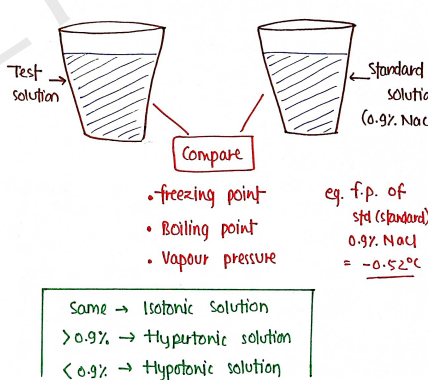
1) Cryoscopic Method : This method is depends upon colligative properties of solution such as freezing point, Boiling point, vapour pressure and temperature difference.
- Take two solution, one standard isotonic solution (0.9% NaCl) and other is test solution (whose tonicity, we have to determine).
- Now Compare their colligative properties with standard solution and determine the tonicity of solution.
2) Hemolytic Method : The effect of various solution of the drug was observed on the appearance of RBC (Red blood cells) suspended in solution.
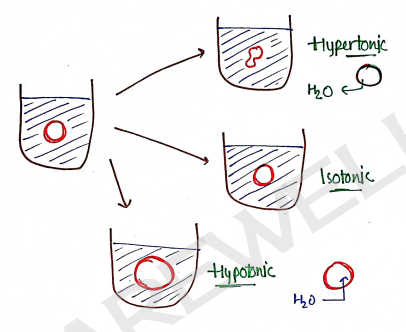
According to osmosis, solvent particle move from low concentration to high concentration.
Hypertonic : Concentration of solution > conch of RBC (0.9%). So, solvent ($H_2O$) move from low to High, RBC to solution, this cause cell shrinkage.
Isotonic : Concentration of solution = conch of RBC (0.9%). So, RBC cells remain constant.
Hypotonic : Concentration of solution < conch of RBC (0.9%). So, Solvent ($H_2O$) move/diffuse from solution To RBC cells, this cause cell swelling.
Method of adjusting tonicity
We can make solution isotonic by using two methods.
- Class I - Cryoscopic & Sodium Chlorid E.
- Class II - White Vincent & Sprowls Method
1. Class I:
- a). Cryoscopic method (freezing point depression method) : This method is used for hypotonic solution, i.e. concentration of solution is less than 0.9% w/v NaCl.
- Sodium chloride (NaCl) is added to solution to make it isotonic.
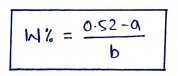
where,
* $w$ = amount of adjusting substance
* $a$ = freezing point of 1% solution of unadjusted solution.
* $b$ = freezing point of 1% solution of adjusting solution.
Sodium Chloride Equivalent (E) : This is used for hypotonic solution, In this add sodium chloride in solution to make it isotonic.

where,
- = Sodium Chloride Equivalent / Amount of NaCl required.
- = constant value
- = Molecular weight of drug solution
2. Class II (White-Vincent / Sprowls method) : This method is used for hypertonic solution i.e. concentration of solution is more than 0.9% w/v NaCl. Water is added to solution to make it isotonic.
- a). White-Vincent method :
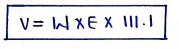
$$V = w \times E \times 111.1$$
where,
* $V$ = volume of water added in solution to make it isotonic
* $w$ = weight of drug in gram
* $E$ = Equivalent weight of drugs (sodium chloride equivalent).
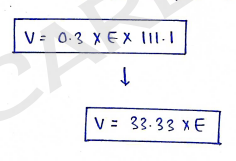
b). Sprowls method : It is the simplification of White and Vincent method, Here weight of drug (w) is set to constant value of 0.3.
Pharmaceutical Importance of Buffers
- Solubility
- Color
- Stability
- Patient comfort
- Optimum pH condition
- Study and research purpose
MAJOR EXTRACELLULAR AND INTRACELLULAR ELECTROLYTES
Chapter-2
Unit-2
SYLLABUS : Major Extra and Intracellular electrolytes: functions of major physiological ions, Electrolytes used in the replacement therapy: Sodium Chloride, Potassium chloride, Calcium gluconate.
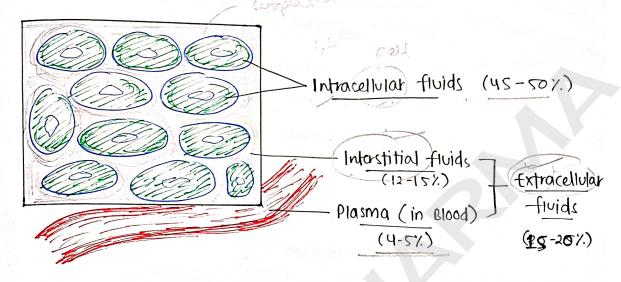
Body fluids
It is a fluids which is present inside human body. About of human body is fluid.
Based on Distribution, fluids are distributed into two types :
- Intracellular fluids - the fluid which present inside the cells
- It is about of body weight.
- Extracellular fluids - the fluid which present outside the cells
- It is about of body weight.
- Interstitial fluids - present between the cells. ($12-15%$).
- Plasma - Inside vascular system (blood) - ($4-5%$).
ELECTROLYTES
It is a substance that ionized when dissolved in suitable ionizing solvent such as water.
- They are also known as ionic solution as they have ions in solution.
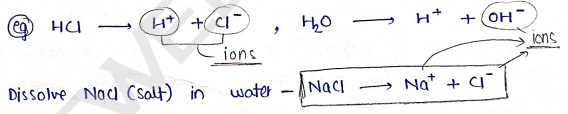
In a solution, an electrolyte is considered to be concentrated if the ions concentration is high; and diluted if the ions concentration is low.
The extracellular fluid contain large amount of ions (eg. etc..) along with nutrients and other substances for the cell (Oxygen, glucose, fatty acids, amino acids etc..).
The intracellular fluid contains large amount of Potassium, Magnesium and Phosphate ions.
FUNCTIONS OF MAJOR PHYSIOLOGICAL IONS
These are those ions/electrolytes present in our body fluids and play major role to maintain homeostasis and keep body fit and healthy.
Major ions/electrolytes :
- Calcium ($Ca^{2+}$)
- Sodium ($Na^+$)
- Potassium ($K^+$)
- Magnesium ($Mg^{2+}$)
- Chloride ($Cl^-$)
- Phosphate
- Bicarbonate ($HCO_3^-$)
I) Calcium ($Ca^{2+}$) : It is important part of teeth and bones, which is also involved in muscle contraction.
- Calcium in higher amount is essential in children for growth and development of bones and tissue.
- Dietary Sources: cheese, green vegetables, eggs and fish.
- Major role:
- In muscle contraction (activation of actin filament)
- Blood coagulation (convert prothrombin to thrombin).
- Formation of Bones and teeth.
- Disease:
- Deficiency (low) of calcium Hypocalcaemia.
- Excess (High) amount of calcium Hypercalcaemia.
II) Sodium ($Na^+$) : It is the main extracellular cation, which is essential for maintaining the osmotic pressure and normal hydration.
- Daily Requirement - about .
- Dietary Sources - Obtained from foods rich in sodium, like salt, buttermilk, cheese, fish, olives, pulses etc..
- It also helps in heart's normal functioning and blood pressure.
- Disease:
- Deficiency of Sodium leads to Hyponatremia.
- Excess level of sodium leads to Hypernatremia.
III) Potassium ($K^+$): It is the major intracellular cation present in the body.
- It is involved in nerve conduction with sodium through Sodium-potassium pump.
- Also helps in pH regulation.
- Daily Requirement - about .
- Dietary Sources - Obtained from foods rich in potassium like apricots, bananas, oranges etc..
- Disease:
- Deficiency of Potassium leads to Hypokalemia.
- Excess amount Hyperkalemia.
IV) Magnesium ($Mg^{2+}$): An important intracellular cation. Around half amount of magnesium ($10-20gm$) is combined with phosphorus and calcium in the bone.
- Daily requirement - about .
- Also helps in glucose metabolism.
- Dietary sources - obtained from nuts, grains, green vegetables.
- Disease:
- Deficiency Hypomagnesemia
- Excess (over) Hypermagnesemia
V) Chloride ($Cl^-$): It is the major extracellular anion. It involve in maintenance of osmotic pressure, proper hydration, and normal cation-anion balance.
- Daily requirement - about .
- Dietary sources - Seaweed, Olives, tomatoes, rye, sea salt and table salt are.
- Diseases:
- Deficiency - Hypochloremia.
- Excess amount Hyperchloremia.
VI) Phosphate ($PO_4^{3-}$): It is the principal intracellular anion, which is present in teeth and bones and also act as a buffer system.
- Daily requirement - about .
- Dietary sources - Obtain from protein rich food, like milk and meat.
- Functions: mild laxative properties, development of teeth and bones, helps in metabolism of glucose & other hexose.
- Diseases:
- Deficiency - Hypophosphatemia
- Excess - Hyperphosphatemia
VII) Bicarbonate ($HCO_3^-$): It is the second most prevalent extracellular anion. It is used as the most important buffer system of the body.
- Maintain acid base balance.
- Metabolic alkalosis and metabolic acidosis may occurs due to deficiency of bicarbonate ions.
- Metabolic acidosis and kidney disturbance are treated with sodium bicarbonate.
ELECTROLYTES REPLACEMENT THERAPY
As we know, electrolytes are important in our body to maintain homeostasis.
Now, sometimes these electrolytes balance is imbalanced due to some condition like Vomiting, Diarrhoea, Dehydration or other disease.
So, Electrolytes replacement therapy is used to maintain/overcome these electrolyte imbalance in which electrolytes are administered externally.
- There are usually three types of compounds/Solutions used in replacement therapy:
- Sodium Chloride ($NaCl$)
- Potassium Chloride ($KCl$)
- Calcium Gluconate ($C_{12}H_{22}O_{14}Ca \cdot H_2O$)
1) Sodium Chloride
- Molecular weight
- Molecular formula
- Method of preparation
- It can be obtained from natural source like sea water and rock salt but in impure form so its purification is necessary.
2. Laboratory method It is prepared by acid-base reaction, in which, strong acid is react with strong base which form sodium chloride salts.
Properties:
- It is colorless, white crystalline solid
- It is odourless
- It is freely soluble in water.
- It is present in crystalline or in saline/salty taste.
- pH neutral.
Chemical reactions:
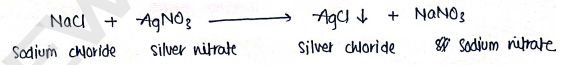
- In this reaction, sodium chloride is reacts with silver nitrate solution, which forms silver chloride precipitate & gives .
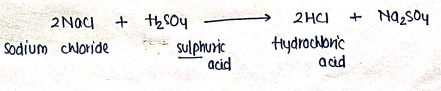
- In this reaction, sodium chloride is reacts with sulphuric acid which forms Hydrochloric acid and give .
Uses:
- Used in treatment of dehydration.
- Used as replacement electrolytes.
Assay of NaCl:
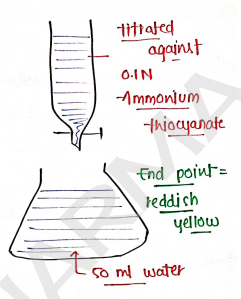
- Take of sample & dissolved in water.
- of
- of Nitric acid and also add Nitrobenzene.
- water.
- Add ferric ammonium sulphate as an Indicator.
- Titrated against Ammonium Thiocyanate till reddish yellow color.
- End point = reddish yellow
- of
2) Potassium Chloride
- Molecular formula:
- Molecular weight: .
Method of preparation:
- When Hydrochloric acid reacts with potassium carbonates it formed potassium chloride. ($K_2CO_3$) $$2HCl + K_2CO_3 \rightarrow 2KCl + H_2O + CO_2 \uparrow$$
- When Hydrochloric acid reacts with potassium bicarbonate ($KHCO_3$) it formed potassium chloride.
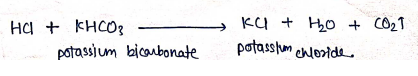
Properties:
- It is crystalline white solid in appearance.
- It is odourless and they have saline/salty taste.
- It is soluble in water but less than and soluble in glycerine and ether.
Chemical Reactions:
- It involves dissociation reaction of potassium chloride into potassium by sodium.
3) Calcium Gluconate
- Molecular formula:
- Molecular weight: .
- It is the calcium salt of gluconic acid containing of calcium ions ($Ca^{2+}$).
Method of preparation :
- When gluconic acid solution is heated with calcium Carbonate (in excess), then filtered & obtained calcium gluconate crystals.
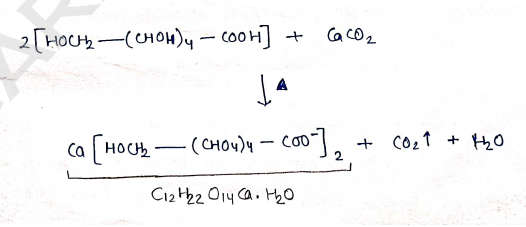
Properties:
- White crystalline powder
- Odourless
- Tasteless
- It is soluble in water and insoluble in alcohol.
- Its solution are neutral to litmus paper.
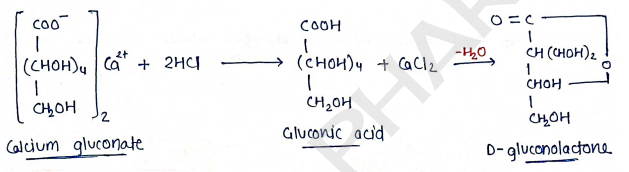
Chemical reactions:
- undergoes decomposition with dilute mineral acids.
Assay:
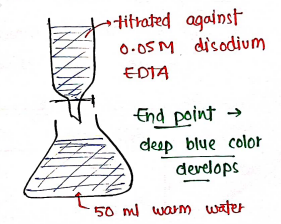
- sample
- of magnesium sulphate
- strong ammonium sol$^n$
- warm water.
- the resultant solution is titrated against disodium EDTA using of mordant black II as an Indicator, until the solution turns deep blue in color.
- End point deep blue color develops
Uses:
- It is used in electrolyte replacement therapy.
- It is also used as a source of .
- Also used for treating the burns caused by hydrofluoric acid.
- Also used to cure mental disorder and mysthenia gravis.
ORS (Oral Rehydration Solutions)
It is simple, cheap and effective and which are used to treat fluid loss.
- It is a solution which restore fluid loss.
- diarrhoea, vomiting or other disease in which body's fluids is decreased.
- ORS provides sodium chloride, potassium, water and other basic nutrients.
- Composition of ORS :
The standard ($310 \text{ mOsm/L}$) ORS formula has been replaced by the new ($245 \text{ mOsm/L}$) formula of ORS as per the recommendation of WHO and UNICEF.
| Composition | Amount | Ions | Concentration |
|---|---|---|---|
| NaCl | |||
| KCl | |||
| Trisodium Citrate | |||
| Glucose | Citrate | ||
| Water | Glucose | ||
| Total Osmolarity |
- In most cases of acute diarrhoea, ($K^+$) ion is lost, so Potassium ($K^+$) is the chief constituents of ion ORS.
- Bicarbonate, Citrate etc are added in ORS to treat acidosis (occurs due to loss of alkali in stools). It promote sodium and water absorption.
Administration:
- Drink ORS at regular intervals of hours.
- In the begining, volume is given in every hours.
Side effects:
- Hypernatremia ($Na^+$)
- Hyperkalaemia ($K^+$)
Uses
- Maintain the level of electrolytes and overcome fluid loss.
ORT - Oral Rehydration Therapy, It is a simple, cheap and effective therapy used for treating dehydration caused by diarrhoea etc..
eg : ORS.
PHYSIOLOGICAL ACID BASE BALANCE
- Our body contain many fluids in which some fluids are acidic and some are basic in nature.
- Now, their acidic/basic nature depends on the pH number which depends on the no. of ions present in fluids.
- Low pH (acidic) High concentration of ions
- High pH (Basic) Low concentration of ions.
The pH level of these fluids is important to perform various function, and if they show large fluctuation, then it may cause severe damage.
- pH of Blood
- Range
- If blood's pH down or up from this range, It may severe damage (also lead to death).
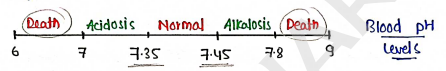
So, it is important to maintain acid-base balance.
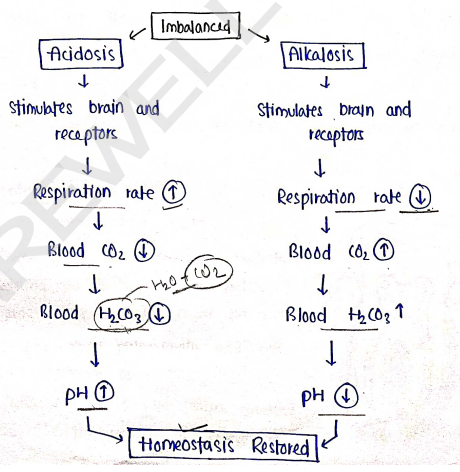
Mechanism of Acid-Base Balance :
- Buffer system
- Renal system
- Respiratory system
I) Buffer System
Buffers are those solution which resist change of pH which maintain acid-base balance. They converts strong acids/bases into weak acid and base.
- Weak acid and its salts.
- Weak base and its salts.
a) Carbonic acid-Bicarbonate Buffer System : In this, Bicarbonate maintain the balance of acid-base by reacting with ions.
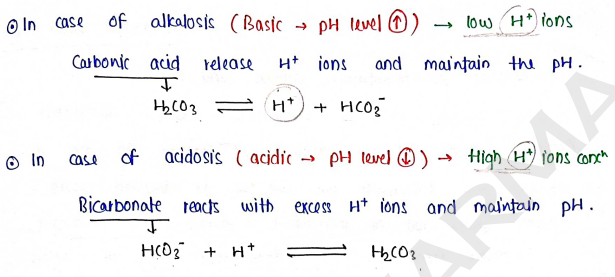
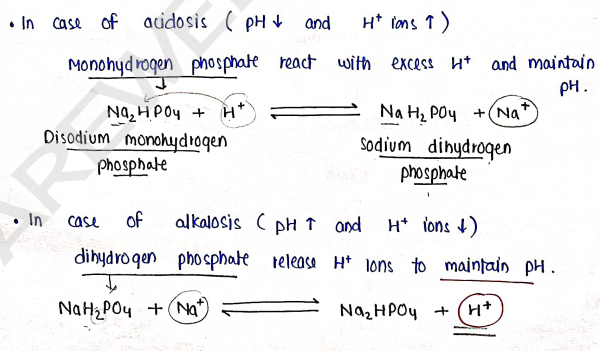
b) Phosphate Buffer System : In this, maintain acid base balance by phosphate. Phosphate present in two form:
- Monohydrogen phosphate ($HPO_4^{2-} / Na_2HPO_4$)
- Dihydrogen phosphate ($H_2PO_4^- / NaH_2PO_4$)
II) Renal System
In this, acid base balance maintained by process of absorption and elimination.
- In case of acidosis (pH level $H^+ \uparrow$) excess amount of ions are eliminated, and the bicarbonate ions ($HCO_3^-$) reabsorbed in our body which helps In the maintainence of acid-base balance.
III) Respiratory system
In which, respiratory system involve to maintain acid-base balance.
DENTAL PRODUCTS
CHAPTER-3
UNIT-2
SYLLABUS : Dentifrices, role of fluoride in the treatment of dental caries, Desensitizing agents, Calcium carbonate, sodium fluoride, and Zinc Eugenol cement.
Dental Products
Dental hygiene is very important which prevent and helps to maintain dental health used in the treatment teeth.
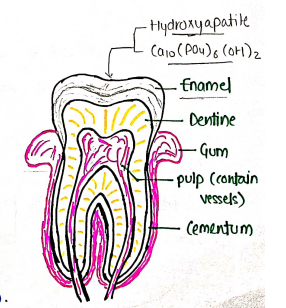
Diagram of Tooth :
- Enamel - It is white hard material hardest part of body. (Above the gum) cover pulp cavity and entire portion of tooth.
- Hydroxyapatite
- Cementum - It is the layer covering the portion of buried tooth.
- Dentine
- Gum
- pulp (contain vessels)
ANTI-CARIES AGENTS
These are those agents which are used in the treatment of dental caries or tooth decay i.e. decalcification of tooth, along with foul mouth odour.
Dental Caries/Tooth decay : It is a dental problem, acids produced by the action of microorganism on Carbohydrates. (which is present on the teeth).

- Fluoride is the main substance, which is used as anti-caries agents.
ROLE OF FLUORIDE IN THE TREATMENT OF DENTAL CARIES
Fluoride is the main substance which is used in the treatment of dental caries / tooth decay.
In human body, fluoride ion is present in trace amount and sufficient quantity is usually obtained from food and water. But where ground water is lacking in fluoride elements, dental problems are more common.
- The process of adding fluoride to the municipal water supply is termed as fluoridation.
Administration : The fluorides can be administered through oral or topical route.
- When fluoride Salt/solution is administered orally/internally, it is rapidly absorbed, transported and deposited in the bone of developing teeth. the remaining conc$^n$ of fluoride is excreted through kidney.
- When fluoride salt/solution is apply topically (on surface of tooth), they prevent dental caries and also avoid the occurence of lesions on the tooth surface due to action of acids/enzyme.
- A small quantity of fluoride (i.e. $1 ppm$) is usually added per day in the preparation, where (more than $2-3 ppm$) more quantity of fluoride may cause mottling of teeth enamel increased bone density, gastric disturbance, muscular weakness and even heart failure.
Mechanism :
- fluorides either work by reducing the ability of bacteria to make acids or by re-mineralising the areas of tooth.
- The fluoride ion ($F^-$) is known to replace the hydroxyl ion ($OH^-$) in hydroxyl apatite, which is main crystalline structure of enamel. The replaced crystal is known as fluorapatite which is more resistant to acids produced by plaque bacteria.

Example
1. Sodium fluoride : ($NaF$) Mol. weight
It is a colorless inorganic compound which is a source of the fluoride ion.
Methods of Preparation :
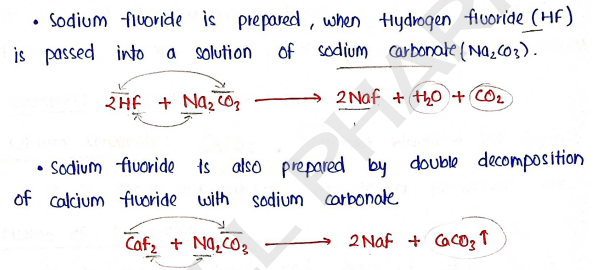
Properties :
- It is colorless/white powder.
- Odourless solid with melting point.
- Soluble in water but insoluble in alcohol.
Uses :
- It is used for oral hygiene for preventing dental caries.
- It makes the teeth enamel resistant to acid, promotes re-mineralisation or reduces production of microbial acids.
DENTIFRICES
It is also part of anticaries agents. It is a substance, which is used for cleaning the reachable surfaces of the teeth with a toothbrush/fingers.
- A dentifrice is used to maintain good oral hygiene.
- It is used for removing dental plaque and food debris.
- Fluorides which are topically effective also present in dentifrices.
Examples: Toothpaste, Tooth Powder, Mouthwash etc..
1). Calcium Carbonate :
Molecular Weight - 100.09 g/mol
It is precipitated calcium carbonate or precipitated chalk.
Method of Preparation:
- It is primarily mined and processed from diverse natural mineral sources.
- It is produced by combining Calcium Oxide ($CaO$) with water to produce Calcium hydroxide ($Ca(OH)_2$), which is then treated with Carbon dioxide to form the Calcium carbonate salt.
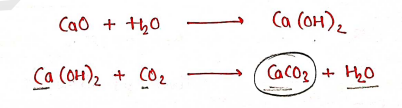
Properties:
- It is white fine powder with melting point .
- It is odourless and tasteless.
- It releases Carbon dioxides ($CO_2$) by reacting with strong acids. $$CaCO_3 + 2HCl \rightarrow CaCl_2 + CO_2 + H_2O$$
Uses:
- It is used in most of the toothpastes and toothpowders as a dental cleaning and polishing agents.
- Also used as inexpensive dietary calcium supplement or antacid.
DESENSITIZING AGENTS
These are those agents which are used to relieve painful sensation in teeth.
- These painful sensation may occurs due to dental caries or when our teeth comes in hot or cold environment.
Example: Silver nitrate, corticosteroids (anti-inflammatory), ($136.28 g/mol$)
It is prepared by heating granulated Zinc ($Zn$) with . $$Zn + 2HCl \rightarrow ZnCl_2 + H_2$$
It is odourless and white crystalline powder.
It is used as desensitizer and also used as antiseptics.
ZINC - EUGENOL CEMENT
- It is a material created by combination of Zinc Oxide and Eugenol present in clove oil.
- It is also known as Zinc Oxide Eugenol ($ZOE$). It is used in dentistry where it is used to fill the cavity of teeth, which occurs due to dental caries.
- This treatment is known as Root Canal therapy ($RCT$).
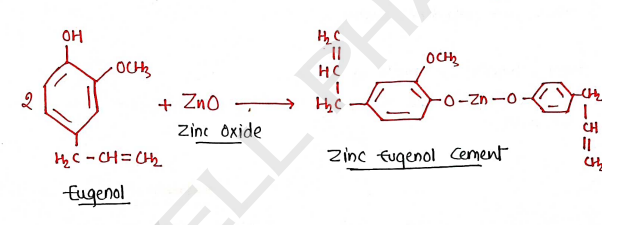
Composition:
- Solid/Powder -
- Zinc oxide (~69.0%)
- Zinc acetate (1.0%)
- Zinc stearate (1.0%)
- White resin (~29.3%)
- Liquids -
- Eugenol (~85%)
- Olive oil (~15%)
Uses :
- In dentistry, it act as filling or cement material.
- In temporary filling for treating dental caries.
Chapter 1

Chapter 2

Chapter 3