Pharmaceutical Inorganic Chemistry - Unit 1
Syllabus
Impurities in pharmaceutical substances: History of Pharmacopoeia, Sources and types of impurities, principle involved in the limit test for Chloride, Sulphate, Iron, Arsenic, Lead and Heavy metals, modified limit test for Chloride and Sulphate
General methods of preparation, assay for the compounds superscripted with asterisk (*), properties and medicinal uses of inorganic compounds belonging to the following classes
Scroll to Download
PHARMACEUTICAL INORGANIC CHEMISTRY
UNIT-1
Syllabus : Impurities in Pharmaceutical Science, History of Pharmacopoeia, Sources and types of Impurities, principle involved in the limit tests for Cloride, Sulphate, Iron, Arsenic, Lead and Heavy metals, Modified limit test for cloride and sulphate.
Pharmaceutical Inorganic Chemistry
It is the branch of Science in which we study about those Inorganic compounds which involves in the preparation of drugs or medicines.
eg: (Acidifiers), Sodium Bicarbonate, Copper Sulphate (Emetics), Antacids
PHARMACOPOEIA
It can be defined as "a book of standards applicable to drugs and their common dosage forms and pharmaceutical aids published in a country under the authority of its own Government."
OR
The term pharmacopoeia comes from the greek word "pharmakon" means "drug" and "poeia" means "to make".
So, It can be defined as "It is a standard official book which contains formulas (monographs) about to make a drugs authorized by Government." It contain a complete details information of drugs.
eg. Name, Method of preparation, Ingredients, etc. IP, B.P, U.S.P etc.
INDIAN PHARMACOPOEIA
It is an official documents responsible for overall quality control and assurance of Pharmaceutical products (pharmaceuticals) in India.
- It defines the identity, purity, safety and strength of drugs.
- This book got approval under the second schedule of the Drug and Cosmetic Act 1940 and Rules 1945.
- The Indian Pharmacopoeia is published by the Indian Pharmacopoeia Commission (IPC) on behalf of Ministry of Health and Family Welfare, Government of India.
- The I.P. Standards are legally acceptable.
History and Development
- In 1833 firstly the East India Company recommended the publication of a pharmacopoeia and Bengal pharmacopoeia.
- From 1885 the B.P. was made official in India.
- After independence, in 1948 the Indian pharmacopoeia committee was developed to publish Indian Pharmacopoeia (IP).
- Now, the first edition of IP was published in 1955 i.e. IP 1955 by IPC.
- It contain Monographs, which contains :-
- List of drugs & related substances
- Sources, tests, formulas
- Uses, dosage and storage conditions etc..
- These information basically used as a reference standard for laboratory, industry and academic institutions.
- Now, editions of Indian Pharmacopoeia was published and several addendum/suppliment are performed :-
INDIAN PHARMACOPOEIA EDITIONS
| EDITIONS | YEAR | ADDENDUM OR SUPPLEMENT | No. of Volumes | MONOGRAPHS |
|---|---|---|---|---|
| 1st edition | 1955 | Supplement 1960 | 2 | 986 |
| 2ND Edition | 1966 | Supplement 1975 | 3 | 890 |
| 3RD Edition | 1985 | Addendum 1989 Addendum 1991 | 2 | 261 |
| 4th Edition | 1996 | Addendum 2000 Addendum 2002 Addendum 2005 | 2 | 1149 |
| 5th Edition | 2007 | Addendum 2008 | 3 | 271 |
| 6th Edition | 2010 | Addendum 2012 | 3 | 52 |
| 7th Edition | 2014 | Addendum 2015 Addendum 2016 | 4 | 577 |
| 8th Edition | 2018 | Addendum 2019 | 4 | 220 |
| 9th Edition | 2022 |
IMPURITIES: SOURCES AND TYPES
- Impurities are Unwanted (foreign) substances, which are present in pharmaceuticals and make it impure.
- Impurities may lowers the quality of pharmaceuticals and also can cause toxicity.
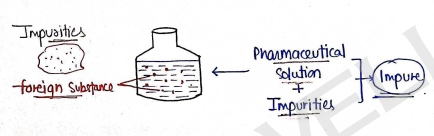
eg : Lead, Calcium, Arsenic, Iron, Chloride etc.
Types of Impurities :
They are mainly of three types
I) Organic Impurities
II) Inorganic Impurities
III) Residual Solvent
i) Organic Impurities : These are those impurities which produced during synthesis of drug substances, and during their storage.
- These impurities are basically "process and drug-related" i.e. it may also arise from raw-material, by-product, excipients, reagents etc..
- These Impurities are not easily identified.
ii) Inorganic Impurities : These are those impurities which basically produced during manufacturing process.
- It can be due to reagents, inorganic salts, heavy metal, catalyst etc..
- These impurities are easily identified by using limit test.
iii) Residual Solvent : These are those impurities which produced during manufacturing process from Solvents.
Impurities may also categorised into two types on the basis of their effects:
- Destructive Impurities : (बिगाड़ना) These are those impurities which basically stop/prohibited the functions of substances.
- It may be removed by chemical method i.e. chemically and physical method i.e. distillation etc.
- Constructive Impurities : (बनाना) These are those impurities which basically improves the functions of substance.
eg: Adding less than 2% carbon in pure iron to manufacture steel.
SOURCES OF IMPURITIES
The impurities in drug products may be obtained from various sources such as during manufacturing, from raw material, excipients or accidentally.
The most common sources are:
i) Raw Materials employed in Manufacture.
ii) Reagents used
iii) Processes or method used
iv) Environment related
v) Defects
vi) Manufacturing Hazards
vii) Storage
viii) Accidental/Adulteration.
(i) Raw Materials : Sometimes, impurities occured from raw material which contaminate the end product.
- (eg) traces of lead, copper, silver etc..
(ii) Reagent used : in manufacturing impurities occurs from reagents if they are not washed properly and contaminate final product.
- (eg) Ammoniated mercury precipitate - impurity.
(iii) Processes : During manufacturing, some impurities enter into the materials.
- It may be due to formulation/excipients.
- also arise due to residue solvent.
- due to chemical process oxidation.
(iv) Environment related : Sometimes, impurities occured from atmosphere, these may cause contamination in final products. Gases - , temp, Humidity etc.
- cross contamination, microbial contamination and packing errors.
(v) Defects : Due to defects occurs during manufacturing such as improper mixing, pressure, pH etc.
(vi) Manufacturing Hazards : Contamination occurs during manufacturing.
- Due to mixing of dirt, glass, metallic or Plastic fragments.
- Due to gross error i.e. Overall error.
(vii) Storage : Sometimes, impurities are occured during storage i.e. dust, insect bodies, animal etc.. may pollute the products.
- eg chemical instability, physical change, temp.
(viii) Accidental : Sometimes, some toxic substances are mixed accidently which we not known.
- eg expensive KBr is adulterated with cheaper NaBr.
Effects :
A little amount of impurity always remain in a material, so if the amount of impurity is present in excess, then it cause toxicity :-
- cause toxic effects.
- alter the physical & chemical properties.
- make therapeutically inactive.
- reduce the Shelf-Life of a substances.
- may changes the color, odour, taste etc and making the substance unhygienic and unfit to use.
LIMIT TESTS
These are quantitative or semi-quantitative test designed to identify and control small quantity of impurities which are likely present in substance.
OR
- These are those tests which are basically used for determination of amount of Impurities. (eg) limit test for chloride, etc.
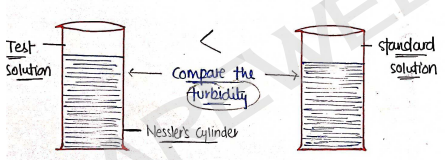
- It is based on the principle that in this, we compare the turbidity/opalescent of test solution with standard solution.
- If the turbidity of test solution is less then standard solution, then limit test passed.
- If, Turbidity of test solution < Turbidity of standard solution Passed
- If, Turbidity of Standard solution < Turbidity of test solution Failed
LIMIT TEST FOR CHLORIDE
It is basically used for identifying the chloride ion impurities and measure their quantity.
- Principle : It is based on the reaction, in which Silver nitrate is react with soluble chloride which form silver chloride which is insoluble in dil. nitric acid precipitates.
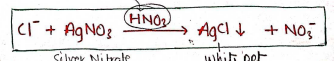
- Now, test solution appears turbidity due to formation of silver chloride (ppt).
- Degree of turbidity is depends on the amount of chloride, which then compared with standard solution.
Procedure : For this, we have to prepare test as well as standard solution.
- Apparatus : Nessler's Cylinder, Measuring Cylinder, Glass rod etc. same for all limit tests.
- Chemicals : Dil. Nitric acid, Silver Nitrate, NaCl.
| TEST SOLUTION (Nessler Cylinder - A) | STANDARD SOLUTION (Nessler Cylinder - B) |
|---|---|
| Specified substance (1g) + 10 ml of water | 1ml of 0.05845% w/v sol^n of sodium chloride |
| + 1ml of Nitric acid ($HNO_3$) | + 1ml of Nitric acid ($HNO_3$) |
| Diluted to 50ml in Nessler Cylinder A | Diluted to 50ml in Nessler Cylinder B |
| + Add 1ml of Silver Nitrate ($AgNO_3$) | + 1ml of Silver Nitrate ($AgNO_3$) |
| set aside for 5 min. | set aside for 5 min. |
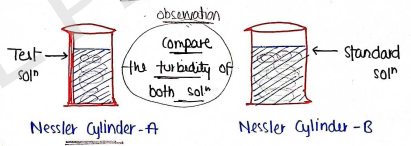
Result :
If the turbidity of test solution is less than the turbidity of standard solution, then sample passed the limit test and ready for use.
If the turbidity of test solution is more than the turbidity of standard solution, then sample failed the limit test and need to control impurities.
Why dil. (Nitric acid) used It is used to make solution (acidic), so they dissolve other impurities except chloride, because they are insoluble in and make it turbid.
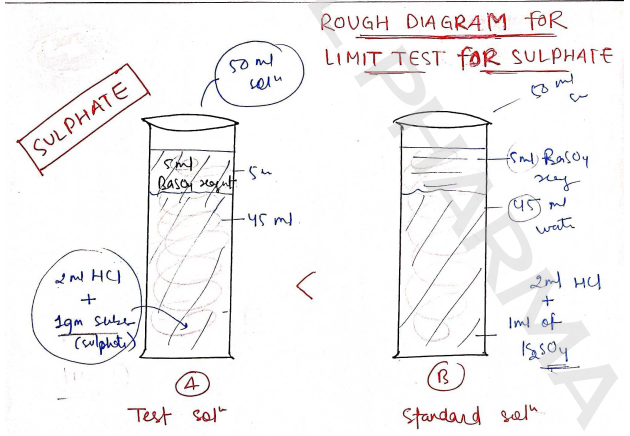
LIMIT TEST FOR SULPHATE
This test is basically used for controlling and identifying the sulphate impurity in substance.
- Principle : It is based on the reaction, in which barium chloride is react with soluble sulphate in the presence of dil. HCl sol^n which forms Barium sulphate precipitate which generate turbidity.
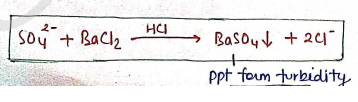
Then this turbidity is compared with std. solution.
In this, Barium chloride ($BaCl_2$) is present in the form of Barium sulphate reagents, which contains, Barium chloride, sulphate-free alcohol, potassium sulphate. (increase the sensitivity of test) as well as prevent super saturation.
Procedure : In this, we have to prepare test as well as standard solution.
| TEST SOLUTION (Nessler Cylinder - A) | STANDARD SOLUTION (Nessler Cylinder - B) |
|---|---|
| 1 gm of specified substance of sulphate | 1ml of 0.1089% w/v sol^n of |
| + 2ml of HCl | + 2ml of HCl |
| Added in 45 ml water | Added in 45 ml dist. water |
| + 5ml of Barium Sulphate reagent | + 5ml of Barium Sulphate reagents |
| Set aside for 5 min | Set aside for 5 min |
| Check the turbidity | Check the turbidity |
Test solution : 1gm of sulphate is weighed and 2ml of HCl is added to 45ml of distilled water, then 5ml of reagent is added to prepare the solution.
Standard solution : 1 ml of 0.1089% w/v solution of is weighed and treated with 2 ml of HCl, then dilute it upto 45ml of water. At last add 5ml of reagent. - [5ml of 0.5M is mixed with 55ml of water, 20ml of sulphate-free alcohol and 5ml of 0.0181% w/v solution of potassium sulphate, then these all diluted upto 100ml water.]
Result : If the turbidity of test solution is less than the turbidity of standard solution, then limit test passed.
MODIFIED LIMIT TESTS FOR CHLORIDE AND SULPHATES
These are those tests which are basically used to overcome those problems which occurs during limit tests.
FOR CHLORIDE
This test is basically used for colored substances, because sometimes we used any colored substance for test solution.
- These colored substance can create problem in formation of turbidity.
- So, In modified limit test, we convert these colored substances into colorless substances, so, we get clear turbidity during test.
eg : (potassium permanganate).
In this, gives purple color which create disturbance in turbidity.
For this, is reacted with ethanol.
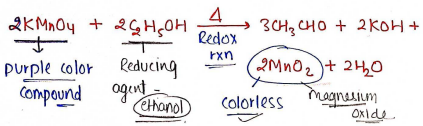
- Preparation of : Dissolve 1.5gm of in 50ml of dist. water, heat on water bath, and add 6ml of 95% ethanol.
- Cool and dilute upto 60 ml.
- then all procedure same as limit test for Chloride.
FOR SULPHATE
- This test is also used for colored substance which create interferance in turbidity i.e. (purple color).
- So, we convert it into colorless substance and follow further procedure of limit test for sulphate.
LIMIT TEST FOR ARSENIC
This test is used for identifying arsenic and controlling Impurities in substance. Arsenic is well known undesirable and harmful substance present in medicine substances.
- Principle : It is based on the reaction, in which firstly arsenic in arseneous acid state is converted into arsine gas by reduction with zinc and hydrochloride acid.
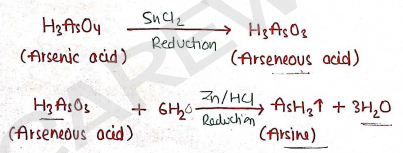
- Then, this arsine gas passed through mercuric chloride paper and produce a yellow stain.
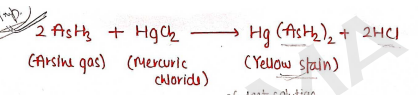
Now, these yellow stain of test solution is compared with yellow stain of standard solution. intensity of these colors.
Apparatus: GUTZEIT APPARATUS
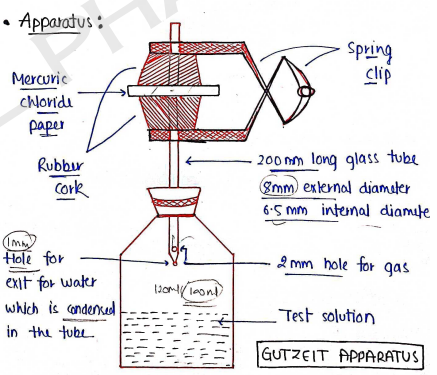
During test, the arsine gas passed through side hole and reached to mercuric cloride paper.
Now, arsine gas produces yellow stain on Mercuric chloride paper.
The intensity of yellow stain is depends on the amount of arsenic impurities present in substances.
Procedure : Prepared both test and standard solution and compared their Intensity of colors.
| TEST SOLUTION | STANDARD SOLUTION |
|---|---|
| Test solution prepared and placed in generator bottle with HCl | 1ml of standard solution of Arsenic with HCl |
| + Add 5ml of 1M KI (potassium iodide)/1gm of KI | + Add 5ml of 1M KI (potassium iodide)/1gm of KI |
| + 5ml of stannous chloride | + 5ml of stannous chloride |
| + 10 gm of Zinc AST (arsenic free) | + 10 gm of Zinc AST (free from arsenic) |
| Keep the solution aside for 40 min. | Keep the solution aside for 40 min. |
| arsine gas react with paper and produce yellow stain | arsine gas react with paper and produce yellow stain |
Compare these intensity/diameter of yellow stain.
If intensity of yellow stain in test solution is less, than limit test passed.
Precautions:
- stain should be compared immediately as it fades away on standing.
- Arsenic chloride paper should be store in dark place.
- the tube should be washed with HCl AST, then with water, then dry it.
LIMIT TEST FOR IRON
This limit test is used for identifying and controlling the iron impurities in solution.
Principle :
It is based on the reaction in which iron is react with thioglycolic acid (mercapto acetic acid) in the presence of ammonium citrate buffer, which forms iron thioglycolate complex of a purple color.
Then the intensity of this purple color is compared with standard solution.
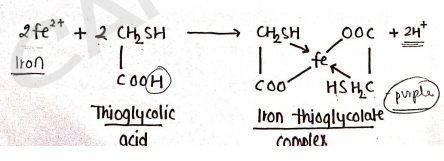
Procedure : Firstly, we have to prepared test as well as standard solution.
| TEST SOLUTION | STANDARD SOLUTION |
|---|---|
| Sample is dissolved in water then volume is made up with 40ml. | 2ml of standard solution of Iron diluted with water upto 40 ml. |
| Add 2ml of 20% w/v citric acid [iron free] | Add 2ml of 20% w/v citric acid [iron free] |
| + | + |
| Add 2 drops of thioglycolic acid (mercapto acetic acid) | Add 2 drops of thioglycolic acid (mercapto acetic acid). |
| Add ammonia to make solution alkaline & make solution upto 50 ml | Add ammonia to make solution alkaline and make solution upto 50 ml. |
| Keep aside for 5 min | Keep aside for 5 min |
| Developed color is compared each other |
- Observation/Result : If the intensity of purple color of test solution is less than standard solution, then sample is passed limit test. In this, the solution should be compared immediately within 5 minutes.
LIMIT TEST FOR LEAD
This limit test is used for identifying and controlling the lead impurities in solution.
Principle :
It is based on the reaction, in which lead is react with diphenylthiocarbazone (dithizone), in alkaline solution to form lead dithizone complex which is red in color.
Dithizone is green in color in chloroform and lead-dithizone complex is violet in color, so the resulting color at the end is red. At last we compare the color.
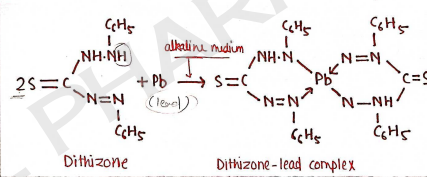
Procedure : In this, firstly we prepared test as well as standard solution, both solutions.
i) Test solution:
- A known quantity of sample solution is transferred in a separating funnel.
- Add 6ml of ammonium citrate (40gm citric acid in 90ml water and ammonia sol^n).
- Add 2ml of potassium cyanide and 2ml of hydroxylamine hydrochloride.
- Add 2 drops of phenol red and make solution alkaline by adding ammonia solution.
- Now, 5ml dithizone solution is used to extract the above solution until it become green.
- Combine the dithizone extracts and transfer to a second separator. (30mg of dithizone in 1000ml of chloroform + 5ml 95% ethanol).
- Shake the combined dithizone extracts for 30 seconds with 30ml of 1% v/v nitric acid solution and discard the chloroform layer.
- Now, in this acid solution add 5ml of standard dithizone solution and also add 4ml of ammonium cyanide. (mg dithizone in 1000ml of chloroform).
- Now, shake the solution for 30 seconds and observe the color of chloroform layer.
ii) Standard solution :
A standard solution of lead is transferred in a separating funnel.
Now, follow the same procedure as test solution.
Observation/Result : The color of chloroform layer developed in test solution should not be more intense than the color obtained in standard solution.
Precaution :
- All reagents/chemicals should be lead free.
- Glassware should washed with dil. or Water.
LIMIT TEST FOR HEAVY METALS
This limit test is basically used for determining the metallic impurities.
- Heavy Metals - these are those chemical elements which have high density and it is toxic or poisonous at low concentration.
examples - Cadmium, Lead ($Pb$), Mercury ($Hg$), Arsenic ($As$), Chromium ($Cr$), Zinc ($Zn$), Bismuth ($Bi$), Cobalt, Silver etc.
Principle : It is based on a reaction in which a solution of heavy metals ($M$) is react with Hydrogen Sulphide ($H_2S$) in an acidic medium, which forms reddish / brown color of metal sulphide.
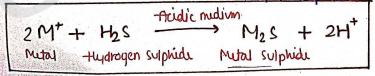
Now, we compare the intensity of color of test solution with standard solution.
In this test, we use lead as standard solution.
It is expressed in 'lead parts per million' i.e. 20ppm for lead test solution should not be more than this.
Procedure :
Prepare test as well as standard solution.
| TEST SOLUTION | STANDARD SOLUTION |
|---|---|
| Specific quantity of sample is dissolved in 25 ml water in 50 ml Nessler cylinder. | Take 2ml of standard lead solution in 25 ml water in 50ml Nessler Cylinder. |
| pH is adjusted between 3-4 by dil. acetic acid/dil. ammonia solution. | pH is adjusted between 3-4 by dil. acetic acid/dil. ammonia solution. |
| volume upto 35 ml water. | Volume upto 35 ml water. |
| Add 10 ml of freshly prepared [Hydrogen Sulphide] | Add 10 ml of freshly prepared [Hydrogen Sulphide] |
| Make volume upto 50ml by adding water. | Make volume upto 50ml by adding water. |
| Keep aside for 5 min | Keep aside for 5 min |
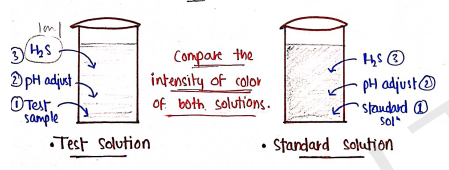
Observation/Result: If the color produced in test solution is less than the color produced in standard, then limit test passed.
It is also performed by other methods :
Method A : for those substances who gives clear colorless solution, used.
Method B : for those substances, which does not give clear colorless solution used.
Method C : for those substances which gives clear colorless solution, used.



